Molar Mass Notes
advertisement

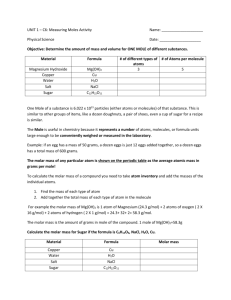
Chemistry Notes: What is Molar Mass? * Remember: atomic mass (g) – mass in grams of 1 mole of the element Molar Mass (MM) = ___grams____ 1 Mol Let’s examine periodic table! If I had one mole of _____ it’s molar mass would be _______ g per 1 mol. Carbon MM= 12.0 g / mol (not diatomic) Atomic Hydrogen MM= 1.0 g / mol Mercury MM= 200.6 g / mol Iron MM=55.8 g / mol Sulfur MM= 32.1 g / mol How many particles do these elements contain? AVOGADROS NUMBER: 1 mol of anything = 6.02 X 1023 atoms How do you calculate the Molar Mass of compounds? You use the formula for that compound. Let’s see! MM= grams/mol EX. Sulfur Trioxide Chemical Formula = _____________ How many total atoms make up one molecule? Atoms = _____________ S: ___ x ______ = O: ___ x ______ = _________ + The molar mass of sulfur trioxide = _______ grams per ________ EX. Phosphorus Trichloride Chemical Formula = __________ How many total atoms make up one molecule? Atoms = _____________ P: ___ x ______ = Cl: ___ x ______ = _________ + EX. Sodium Hydrogen Carbonate Chemical Formula = __________ How many total atoms make up one molecule? Atoms = _____________ Na: ___ x ______ = H: ___ x ______ = C: ___ x ______ = O: ___ x ______ = _________ + Covalent Compounds are made up of _____________. Ionic Compounds are made up of _______________. Elements are made up of _____________. Now let’s use Molar Mass to find other measurements! MOLE Mass Conversions And MASS Mole Conversions Use your Road Map! Book pg. 298 Ex. #1 Write the chemical formula for aluminum oxide ___________ What is given? _________ Trying to find? ____________ ______________ x _______________ = g of Ex. #17 Write the chemical formula for iron (II) hydroxide ___________ What is given? _________ Trying to find? ____________ ______________ x _______________ = g of Ex. #18 Write the chemical formula for boron ___________ What is given? _________ Trying to find? ____________ ______________ x _______________ = g of VOLUME Mass Conversions And VOLUME Mole Conversions Use our Mole Road Map! Book pg. 301 At STP (Standard Temp and Pressure): Get out Reference Table pg. 1 1 mole of gas = 22.4 L of gas = 6.02 x 1023 particles of gas Problem 10.7 What is the correct formula for sulfur dioxide? _____ What is given? ___________ Trying to find? _________ ______________ x _______________ = Problem 10.8 pg. 302 What is given? __________ L of Density = __________ = ________ Trying to find? ___________ #18 HINT: Density = __________ = ________ What is given? __________ Trying to find? ___________