SOP for UV-Vis and Fluorescence
By Dan Palumbo and EJ Habina
Introduction
A UV-Vis spectrophotometer is an instrument commonly used in lab that analyzes compounds in
ultraviolet and visible regions of electromagnetic spectrum. Unlike infrared which looks at vibrations,
UV-Vis looks at electronic transitions. The UV-Vis instrument allows for the determination of
wavelength and maximum absorbance of compounds.
The Beer-Lambert Law, A = εbc, where A is Absorbance, ε is molar extinction, b is path length,
and c is concentration, directly applies to the UV-Vis instrument.
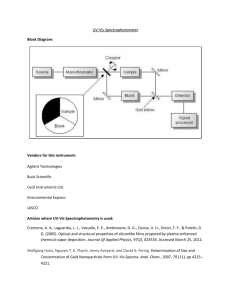
The instrument used is the Thermo Evolution 600 UV-Vis spectrophotometer. “The optical
design of the Evo 600 includes dual deuterium and tungsten sources and a high performance
photomuliplier detector. Its features provide enhanced high-level performance for the Material Science
market and general research applications”
(http://www.thermo.com.cn/Resources/200802/productPDF_1838.pdf).
While UV-Vis has limits in detection, fluorescence can find trace concentrations as low at 1 ppt.
Fluorescence uses two wavelengths along the spectrum to determine the quantitative measurements of an
analyte. The fluorescence instruments send light into a sample and excite the electrons in a molecule. The
instruments then read the radiation released when the electrons return to their ground states.
The instruments used are the Shimadzu Spectrofluorophotometer and the PE Luminescence
Spectrophotometer.
Purpose of the instrument
As previously stated, the UV-Vis instrument allows for the determination of wavelength and
maximum absorbance of compounds. The instrument has a wide variety of uses including life science
biochemical, pharmaceutical and quality control, and material science. It can be used in nucleic acid tests,
to test for protein concentration, thermal denaturation/renaturation, and kinetics.
The fluorescence instrument is a type of electromagnetic spectroscopy which analyzes
fluorescence from a sample. Fluorescence spectroscopy is used in biochemical, medical, and chemical
research fields for analyzing organic compounds.
Experimental Purpose
UV-Vis:
The purpose of this lab is to determine the absorbance levels of Salicylic Acid in aged aspirin
samples by running samples of SA in hexanes through the UV-Vis instrument.
Fluorescence:
The purpose of the next part in the lab is to determine the amount of quinine in tonic water
through the fluorescence instruments.
Experimental Procedure
UV-Vis:
1. Prepare standard solutions of 40, 25, 10, 7 and 2ppm from a 1000ppm stock solution of Salicylic
Acid (SA) in hexanes. Use 50mL volumetric flasks to dilute the stock solution with hexane
2. Run 3 samples of each standard through the UV-Vis spectrometer
3. Create calibration curves from the data returned from the UV/vis
4. Dissolve and filter an aspirin tablet through hexane. Run this new solution for SA absorbance on
the UV-Vis instrument
Fluorescence:
1. Prepare standard solutions of 2ppm,3ppm, 4ppm, 5ppm solutions of quinine in .1M H2SO4 using
the 1000ppm stock solution
2. Run 3 samples of each concentration through the fluorescence instruments and record the
wavelength and intensity of the largest peak
3. Make a solution of tonic water by diluting 5mL of tonic water with 95mL of H2SO4 and run 3
scans of the solution through the fluorescence instruments. Record the wavelength and intensity
of the largest peak
Instrument Procedures
UV-Vis:
1. Power on the instrument by pressing the switch on the left side near the power cord
2. Turn on the monitor and double-click ‘VisionPro’. If the computer is not logged in, use the
Username: student, with no password. Enter it again if it does not log in the first try
3. Instrument will run through a startup check for about 10-15 minutes
4. In the toolbar frame, select the Scan icon (looks like a scatter plot with curved lines)
5. In the dialog box, enter the wavelength from 190-400nm, choose Absorbance, and Tungsten
Lamp
6. Click on Baseline Correction and make sure 100%T is selected
7. Next click on the Baseline/Zero icon (looks like a graph with arrows pointing down)
8. Wash a quartz cuvette with hexane 3 times, then fill 3/4th of the way and dry with kim wipes.
Place the cuvette in the first slot with the frosted sides facing you and the window
9. Next click on the Run icon (looks like a man running)
10. To obtain peak and abs, click on the graph window, then go to the graph menu at the top and
choose Peak Labels and select Wavelength and Value. In the menu graph again select Track at
the top of the list and click on Graph. Using the arrow keys, find the peak around 310nm and
record the abs value
11. Repeat steps 8-10 with each standard and aspirin solution
Fluorescence:
1. Shimadzu:
a. Turn on the instrument and computer
b. Open the PX-150x program
c. Before starting anything, check the lower right corner and see if the indication says the
instrument is on. If not:
i. Go to the Configure menu and go down to the PC Configuration and make sure
the First Portal is selected for the instrument
ii. Then also under the Configure menu, go to the Instrument and make sure that On
is selected
d. Set the instrument to Quantitative Analysis by going to Acquire Mode and selecting
Spectrum
e. Obtain stock solution of 1000ppm quinine in .1M H2SO4. With the stock solution make
2ppm,3ppm, 4ppm, 5ppm solutions of quinine in .1M H2SO4
f.
Fill a cuvette with some 2ppm solution, run the instrument under these settings
i. Spectrum Type: Excitation
ii. EM Wavelength: 400nm
iii. Ex Wavelength Range: 220 (start); 900 (end)
iv. Sensitivity: Low
v. Scan Speed: Fast
vi. Recording Range: -10.00 (low); 500.00 (high)
vii. EX/EM Slit Width: 10/10
viii. Response Time: 0.02
ix. Repeat Scan/ Auto File: No setting needed
g. Then click Search λ and click Search. Ensure that the default range for the
EX(Excitation) and EM(Emission) are 230-450nm and 240-650nm respectively. Once
the optimal wavelengths are found for EX and EM, note them. Then reset the parameters
to be:
i. Spectrum Type: Emission
ii. EX Wavelength: The ideal wavelength found in the optimal wavelength search
iii. EM Wavelength Range: Make sure that the optimal EM wavelength is included
in the range
iv. Sensitivity: Low
v. Scan Speed: Fast
vi. Recording Range: -10.00 (low); 500.00 (high)
vii. EX/EM Slit Width: 10/10
viii. Response Time: 0.02
ix. Repeat Scan/ Auto File: No setting needed
h. Run a scan of each concentration (2ppm, 3ppm, 4ppm, and 5ppm) and record the
wavelength and intensity of the largest peak. Repeat 3 times for each
i.
Make a solution of tonic water by diluting 5mL of tonic water with 95mL of H2SO4
j.
Run a scan of this solution and record wavelength and intensity of largest peak. Repeat 3
times
2. Perkin Elmer:
a. Turn on instrument and computer
b. Open the WINLab program
c. Fill a cuvette with your blank (H2SO4) and run a scan with the same parameters as before
except change the slit from 8 to 3 for both
d. Run your samples from the other fluorescence instrument each three times. Record the
wavelength and intensity of the largest peak. To find these numbers, click the button with
a peak with a vertical line through it and drag the line to the middle of the largest peak.
The wavelength and intensity are found towards the bottom of the screen
e. Repeat for your tonic sample 3 times
 0
0