UV/Vis and Fluorescence
advertisement

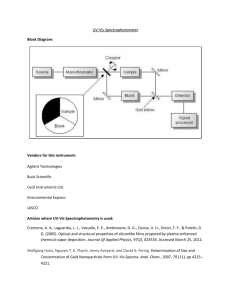
UV-Vis and Fluorescence Purpose: The purpose of this lab is to learn and become familiar with the UV-Vis and fluorescence. The UV-Vis will test aspirin samples and the fluorescence will test quinone and sulfuric acid. Introduction: Ultraviolet and visible spectroscopy is commonly used for the quantitative determination of a large number of inorganic, organic, and biological species. UV-Vis is based on the measurement of transmittance or absorbance of solutions contained in transparent cells having a path length in centimeters. The instrument consists of a source to provide energy, a filter or monochromator, a reference cell, a photo-detector, amplifier, and readout. Fluorescence is used for the quantitative determination of large elements that specifically form vapors and hydrides. Fluorescence instruments are complex, expensive, and less sensitive. Instrumentation consists of a source and wavelength selector set at a right angle to the sample, a second wavelength selector, detector, and finally a signal processor and readout. Procedure: UV-Vis Thermo Evolution 600 UV-Vis Turn on Instrument and open the “VisionPro” program on the adjacent computer The instrument will perform a startup check to see that everything is in working order Select the Scan icon on the tool bar. Four viewing windows should be on screen, in the top center one make the following adjustments: o Wavelength: 190nm-400nm o Scanning for: Absorbance o Lamp: Tungsten Set the Baseline Correction for 100%T Prepare Blanks: Wash a quartz cuvette with hexane multiple times, fill 3/4th full, and dry with Kim wipes Place one blank in each sample holder After setting the Baseline Correction the program will have had a request for blanks, and a simple approval of the request will start the baseline scan, if denied, simply press baseline/zero icon In the same window from step 5s adjustments; set the results dropdown menu to peak pick Prepare sample/standard the same way as step seven and place in the front holder and leave blank in the back one Press the Run icon (appears to be a person running) Run each sample/standard three times without opening the instrument Repeat steps 10-12 for remaining sample/standards Click on sample name on left and right click. Click plot sample. This puts all samples on one graph. When finished: Close out program, turn off UV-Vis, turn off computer monitor [LEAVE COMPUTER ITSELF ON] Fluorescence: Shimadzu: Turn on the instrument (bottom right) and computer Open the PX-150x program Check lower right corner to see if the indication says the instrument is on. Go to Configure > go down to the PC Configuration > make sure the First Portal is selected for the instrument Configure menu > go to the Instrument and make sure that On is selected Set the instrument to Quantitative Analysis by going to Acquire Mode > selecting Spectrum With the 1000 ppm stock solution, make a 100 ppm stock solution Make 2 ppm, 3 ppm, 4 ppm, 5 ppm standards of quinine in 0.1M H2SO4 Fill a cuvette with some 2ppm solution, run the instrument under these settings: Spectrum Type: Excitation EM Wavelength: 400nm Ex Wavelength Range: 35o nm to wavelength that contains peak Sensitivity: Low Scan Speed: Fast Recording Range: -10.00 to an amount that shows entire peak EX/EM Slit Width: 10/10 Response Time: 0.02 Repeat Scan/ Auto File: No setting needed/ disable Then click Search λ and click Search. Ensure that the default range for the EX (Excitation) and EM (Emission) are 350 - 600 nm and 240 - 650nm respectively. Record optimal wavelengths found for EX and EM. Spectrum Type: Emission EX Wavelength: The ideal wavelength found in the optimal wavelength search EM Wavelength Range: Make sure that the optimal EM wavelength is included in the range Sensitivity: Low Scan Speed: Fast Recording Range: -10.00 (low); 500.00 (high) EX/EM Slit Width: 10/10 Response Time: 0.02 Repeat Scan/ Auto File: No setting needed/disable Run a scan of each concentration and record the wavelength and intensity of the largest peak. Repeat 3 times for each. Repeat for sample. Data: All graphs are in Instrumental Notebook. Standard Calculations: 1000 𝑝𝑝𝑚 = 0.1 𝑆𝑢𝑙𝑓𝑢𝑟𝑖𝑐 𝐴𝑐𝑖𝑑 100 𝑚𝐿 (5 𝑝𝑝𝑚)(100 𝑚𝐿) = (1000 𝑝𝑝𝑚)(𝑥 𝑚𝐿) = 0.5 𝑚𝐿 (10 𝑝𝑝𝑚)(100 𝑚𝐿) = (1000 𝑝𝑝𝑚)(𝑥 𝑚𝐿) = 1.0 𝑚𝐿 (1 𝑝𝑝𝑚)(100 𝑚𝐿) = (1000 𝑝𝑝𝑚)(𝑥 𝑚𝐿) = 0.1 𝑚𝐿 (. 2 𝑝𝑝𝑚)(100 𝑚𝐿) = (1000 𝑝𝑝𝑚)(𝑥 𝑚𝐿) = 0.02 𝑚𝐿 Calibration Curve: UV-Vis Calibration of UV/Vis Absorbance 0.1 y = -0.0769x + 0.0827 R² = 0.9979 0.08 0.06 0.04 0.02 0 0 0.2 0.4 0.6 0.8 1 1.2 Concentration (ppm) Calibration Curve: Fluorescence Absorbance Calibration of Fluorescence 1000 800 600 400 200 0 y = 163.51x + 21.647 R² = 0.9998 0 1 2 3 4 Concentration (ppm) Conclusion: 5 6 Overall, the UV-Vis and fluorescence were interesting instruments to learn. Both instruments were easy enough to use. The initial concentrations were a little too strong for the instrument so we had to dilute them to a much smaller concentration, none higher than 2 ppm. The samples used were filtered and unfiltered and provided very similar results each time. The data from the fluorescence was all very similar to each other, with the variations being between the 200 and 240 nm range, as the concentrations got greater. Error was found in our inexperience with the instrument.