UV- Vis and Fluorescence Lab
advertisement

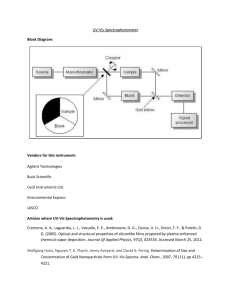
Brittany Hicks UV-VIS & Fluorescence Lab- Dr. Foy Spring 2013 UV-VIS and Fluorescence Introduction The ultraviolet-visible spectrophotometer is a chemical instrument that analyzes compounds that fall under the ultraviolet and visible light spectrum regions of the electromagnetic spectrum. The goal of this instrument is to analyze compounds through electronic transitions. Wavelength and maximum absorbance can both be determined. Determination of particular concentrations of the sample can be explained through Beer’s Law. Analytical analysis for the determination of different analytes is primarily used in the UV-VIS, in which absorbance and wavelengths are optimal comparisons. Fluorescence is very similar in technique and analysis of the UV-VIS. The main difference between the two instruments is that fluorescence uses atomic emission rather than absorption. Fluorescence uses a beam of light that excites electrons in molecules of certain compounds. These compounds then emit a wavelength of light. Electromagnetic spectroscopy is used in many different analysis of medical, chemistry and biology departments. The purpose of this experiment was to familiarize ourselves with both of these instruments so that proper analysis could take place. Aspirin was the main sample under analysis. Using the UV-VIS, aspirin was tested to determine the amount of salicylic acid. Fluorescence will analyze aspirin using quinine and tonic water to determine amounts. Both known and unknown samples were both ran. Procedure - Spectrofluorophotometer Turn on instrument in the bottom right hand corner, and also the computer Open the PX-150x program Check lower right corner of screen to see if indications says instrument is on o If not, go to configure > PC configuration > select the First Portal for the instrument o Back to Configure menu > Instrument > On Go to Acquire mode > Spectrum Place a cuvette of H2SO4 in the instrument o Configure > Parameters > Spectrum type: excitation o EX wavelength: 400 nm o EM wavelength: 220-900nm o Sensitivity: Low o Scan Speed: Fast o Recording Range: -10-500 (may need to be adjusted) o EX/EM slit width: 10/10 o Response time: .02 o Repeat Scan/Auto File: no setting needed o Click Ok Place cuvette of smallest ppm in the instrument o Click Search λ icon in bottom right of screen o Set EX to around 230-450 and EM to 230-650 o Click search o Record optimal wavelengths for EX and EM Go to Parameters > Spectrum type > Emission o EX wavelength: optimal wavelength from search o EM wavelength: range needs to include optimal wavelength o Sensitivity: Low o Scan Speed: Fast o Recording Range: -10-500 o EX/EM slit width: 10/10 o Response time: .02 o Repeat Scan/Auto File: No setting needed Run a scan of each concentration 3 times, recording the wavelength and intensity of the highest peak for each trial Run tonic water dilution as unknown also repeating 3 times and recording wavelength and intensity of the highest peak Procedure – UV-VIS Turn on UV-Vis Open Vision Pro Software Wait for the set up scan to complete Once that is done, select the scan icon on the toolbar o Set wavelength to Start:190 Stop:400 o Scan for: Absorbance o Lamp: Tungsten Go to Set-up > Baseline tab > 100% T To blank the instrument fill a clean cuvette ¾ with hexane and place in front slot o Click the baseline zero icon When that is complete, remove the blank and put the first sample in Click the run icon Run each sample three times only opening the instrument to put new sample in When finished, close out computer program and turn off instrument Data and Analysis Unfortunately chromatograms could not be obtained for this lab from the UV-VIS instrument. However, all the data was written down in my lab notebook before the program was shut down. A hand drawn picture of the chromatograph showing how the peaks looked for each concentration as well as where the unknown sample was pictured in the overall figure. Both the quantitative data and figure of concentration graph are attached within the lab notebook. UV-VIS DATA Concentrations Prepared 10ppm 20ppm 30ppm 40ppm 50ppm 100ppm Aspirin Sample Absorbance 1 Absorbance 2 Absorbance 3 0.147 0.157 0.210 0.259 0.273 0.716 0.223 0.136 0.182 Concentration Run 1 wavelength 0.1ppm 466 Run 1 intensity 30.25 Run 2 wavelength 466 Run 2 intensity 28.80 Run 3 wavelength 466 Run 3 intensity 28.60 1.0ppm 466 265.85 466 774.40 466 767.24 5.0ppm 467 1015.69 467 1015.66 467 1015.62 0.171 0.419 0.696 0.789 1.545 0.786 0.277 0.239 0.383 0.451 1.345 0.442 Fluorescence Data 10.0ppm MAX MAX MAX Blank 360 5.73 360 5.27 360 5.47 Unknown 467 876.41 467 858.84 467 841.98 ***Optimal Wavelength… Excitation= 344 Emission= 449 1200 1000 800 600 400 200 0 0 1 2 3 4 5 6 7 This graph shows concentrations verse the intensities received from Fluorescence procedure Conclusion Both instruments were used pretty easily, except for the UV-Vis closing out of the program before chromatograms could be saved as a picture file. All of the data looks good overall in comparison to results other classmates received on these instruments as well. There are minor differences in results which could have possibly been to dirty cuvettes being analyzed containing the samples. More concentrations could have been made up for both of these instruments for further testing and to see if better results could have been obtained. Our regression looking at the concentration vs. intensities was not very good which could be fixed by running more samples and concentrations.