HW chapter 1
advertisement

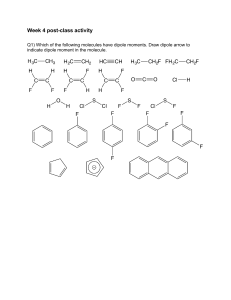
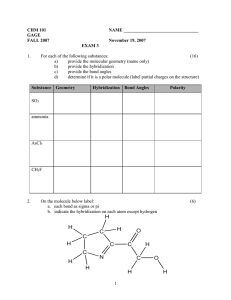
CHM 201 HW chapter 1 Name Due Thursday 4/11/13 *Show your work clearly and staple your papers OR you will get ZERO 1) Consider hydrogen cyanide, a) What is the hybridization at the N? at the C? b) What are the types of three CN bonds? What orbitals are overlapping to form them? (E.g sp3C-sp3N ) c) In what type of orbital are the unshared electrons on the N? d) Draw the bonding picture showing how the orbitals overlap to form the pi bond 2) Show a Lewis structure for cyanate anion (OCN-). Which atom has the negative formal charge in your structure? Show its resonance structure and rank them 3) What is the hybridization and geometry angle at all atoms, except hydrogens, in these compounds? a. b. c. d. 4) Use heavy and dashed wedge lines to show the shapes of the following molecules. Show the bond dipole of each polar bond and show the overall dipole of each molecule. a. b.