Vasquez High School -- AP Chemistry B -- Test #2b -
advertisement

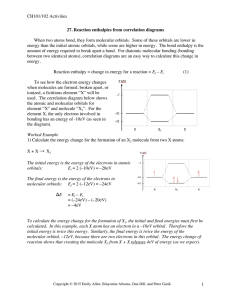
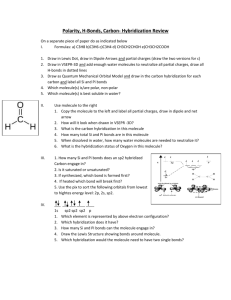
Vasquez High School -- AP Chemistry B -- Test #2b -- Chapter 10 -- 50 points Write TRUE if the statement is true OR write the word(s) that substitutes for the underlined word(s) that would make it true. Writing false earns partial credit. Three points each. _______________ 1) A common yet very dangerous gas used in the semiconductor industry is silane, SiH4. A molecule of silane most likely has a octahedral shape. 2 _______________ 2) The sp hybrid bond orbital is of lower energy than the normal 2p orbital. _______________ 3) In sp3d2 hybridization, the six hybrid orbitals are directed toward the corners of a(n) octahedron. _______________ 4) Molecules are stable if the number of electrons in bonding molecular orbitals is greater than the number in antibonding molecular orbitals. _______________ 5) The dipole moment is found by multiplying charge by time. Short Answer/Fill-in/Diagram section. Be neat and complete. Five points each. 6) Draw the Lewis structure of a benzene molecule at the right. Then describe the nature of their pi bonds and why this is so. ________________________________________________ ________________________________________________ ________________________________________________ ________________________________________________ ________________________________________________ 7) Define how bond order is determined and then show, using it, how the proposed He2 cannot be stable. 8) Consider the molecule propyne, C3H4. Draw the Lewis structure. Describe how many sigma bonds and pi bonds are present. For three points each, predict the geometry of each of the following: 9) ZnCl4- 10) AsBr5 11) H2S Three points each. 12) VSEPR stands for _____________________________________________________________________. 13) The shape of the ammonia molecule, NH3, is ________________________________________________. 14) For five points, list the five most important hybrid orbital types, that is, hybridization of the central atom, and the resulting shapes of molecules containing them: _______________ _________________________________________________ _______________ _________________________________________________ _______________ _________________________________________________ _______________ _________________________________________________ _______________ _________________________________________________