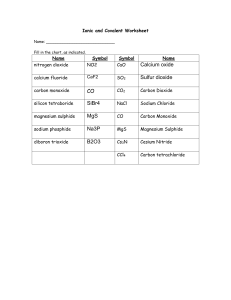
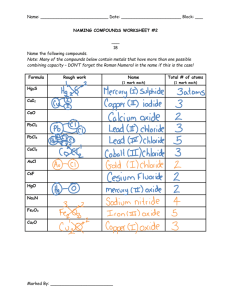
Binary Compounds Formula Writing
advertisement

NAME_______________________________________________ PERIOD ____ BINARY COMPOUNDS : FORMULA WRITING 1. look up the elements represented in the name and find their oxidation numbers (charges) 2. How Many? Multiply by the number of pieces (atoms) that will make the total + and _ charges match. 3. The number you multiplied by in step #2 IS the subscript for the formula SEE EXAMPLES Elements Final formula Ions & Oxidation # HOW TO SHOW WORK Ex 1. potassium bromide K+1 Br -1 K1Br1 OR KBr +1 x 1=+1 -1 x 1 = -1 +1 Ex 2. sodium phosphide Na3P1 OR Na3P Na P-3 +1 x 3 = +3 1. lithium fluoride 2. beryllium bromide 3. potassium iodide 4. magnesium sulfide 5. calcium nitride 6. hydrogen selenide 7. potassium oxide 8. aluminum bromide 9. zinc phosphide 10. silver oxide 11. gallium chloride 12. barium nitride 13. strontium fluoride 14. beryllium sulfide 15. calcium nitride 16. magnesium phosphide 17. potassium iodide 18. sodium oxide 19. zinc chloride 20. lithium phosphide -3 x 1 = -3