How to Balance Ionic Compounds Wksht
advertisement

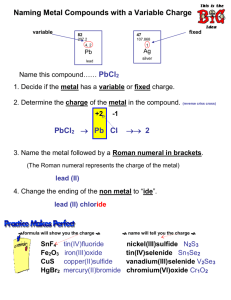
How to Balance Compounds Step 1: Put charges above each ion in the compound. (ex: Ca+2OH-1) Step 2: Criss Cross Method: Balancing out compounds means to balance out the charge of the compound so that the net charge is ZERO. (ex: Ca+2(OH-1)2) Calcium has a +2 charge and hydroxide has 2 -1 charges, when added together, the net charge is zero. Notice that we had to put a parenthesis around t he hydroxide so that the O and the H both have the quantity of two. Practice Problems: 1. CdF = 2. CeS 3. RbO 4. MgS 5. NiN 6. BiN 7. CaI 8. Pb+2S 9. NH4Cl 10. CaBrO3 Worksheet-Monatomics Directions: Write balanced formulas. (Write ions, place charges above, and balance!!) 1. Sodium Bromide ______________ 33. Cobalt (II) Fluoride ___________________ 2. Barium Sulfide _______________34. Manganese (III) Sulfide ___________________ 3. Iron (III) Phosphide _______________35. Silicon Nitride ___________________ 4. Copper (II) Iodide _______________36. Cobalt (III) Selenide ___________________ 5. Iron (II) Bromide _______________37. Lead (IV) Chloride ___________________ 6. Magnesium Boride _______________38. Manganese (III) Bromide 7. Silicon Oxide _______________39. Aluminum Selenide _________________ 8. Bismuth Fluoride _______________40. Iron (II) Hydride _________________ 9. Potassium Nitride _______________41. Cerium Oxide _________________ 10. Tin (II) Fluoride _______________42. Cobalt (III) Boride _________________ 11. Tin (IV) Sulfide _______________43. Chromium (II) Selenide _________________ 12. Tin (II) Hydride _______________44. Manganese (II) Iodide _________________ 13. Copper (I) Boride ___________________ 14. Silver Sulfide ___________________ 15. Lead (IV) Iodide ___________________ 16. Chromium (III) Bromide ___________________ 17. Rubidium Oxide ___________________ 18. Cesium Chloride ___________________ 19. Cerium Sulfide ___________________ 20. Mercury (II) Iodide ___________________ 21. Lead (II) Chloride ___________________ 22. Cobalt (II) Fluoride ___________________ 23. Iron (III) Sulfide ___________________ 24. Strontium Fluoride ___________________ 25. Aluminum Boride ___________________ 26. Lithium Nitride ___________________ 27. Cadmium Boride ___________________ 28. Manganese (II) Iodide ___________________ 29. Cobalt (III) Selenide ___________________ 30. Tin (IV) Phosphide ___________________ 31. Iron (III) nitride ___________________ 32. Chromium (III) Fluoride ___________________ __________________