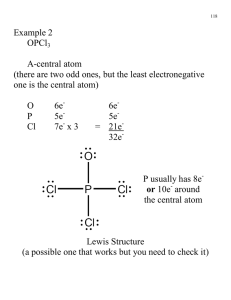
Molecular Orbital Theory
advertisement

Molecular Orbital Theory Chemistry 123 Dr. Patrick Woodward Supplemental Lecture 2 Atomic Orbitals Think of the electrons in atomic orbitals as standing waves. The color coding gives the phase of the wave. When the wavefunction is colored blue the wave is positive, when it is colored red it is negative. See: http://winter.group.shef.ac.uk/orbitron/ for a visual depiction 1 Molecular Orbital (MO) Theory MO theory is based on the premise that electrons in molecules reside in molecular orbitals. These molecular orbitals are formed from the overlap of atomic orbitals. MO’s can be classified as bonding, antibonding and nonbonding. Bonding Molecular Orbital • Constructive Interference • Increases electron density between nuclei • Forms a bond Antibonding MO • Destructive Interference • Decreases electron density between nuclei (nodal plane) • Cancels a bond Image taken from http://www.sparknotes.com/chemistry/bonding/molecularorbital/section1.html MO Diagram for H2 Antibonding MO Bonding MO Image taken from http://www.sparknotes.com/chemistry/bonding/molecularorbital/section1.html 2 Energy level diagrams / molecular orbital diagrams Single (sigma) bond between H atoms No bond between He atoms Fundamental Principles of MO Theory Molecular Orbitals (MO’s) from Atomic Orbitals (AO’s) 1. # of Molecular Orbitals = # of Atomic Orbitals 2. The number of electrons occupying the molecular orbitals is equal to the sum of the valence electrons on the constituent atoms. 3. When filling MO’ MO’s the Pauli Exclusion Principle Applies (2 electrons per Molecular Orbital) 4. For degenerate MO’ MO’s, Hund's rule applies (fill each with a single electron before pairing up 2 electrons in a single orbital). 5. AO’ AO’s of similar energy combine more readily than ones of different energy 6. The more overlap between AOs the lower the energy of the bonding orbital they create and the higher the energy of the antibonding orbital. 3 MOs from 2p atomic orbitals sigma antibonding (σ∗) pi antibonding (π∗) sigma bonding (σ) pi bonding (π) 1 sigma interaction: Through overlap of orbitals along the internuclear axis. 2 pi interactions: Through overlap of orbitals above and below (or to the sides) of the internuclear axis (nodal plane – zero overlap along the internuclear axis). MO Diagram for a second row diatomic molecule (i.e. O2) 4 Metal d – ligand p MO’s M dz2 – L p σ* M dx2-y2 – L p σ* Ligand p orbitals Metal d orbital M dxz – L p π* M dxy – L p π* M dyz – L p π* 5