Diatomic Molecule MO Diagrams: Chemistry Worksheet
advertisement
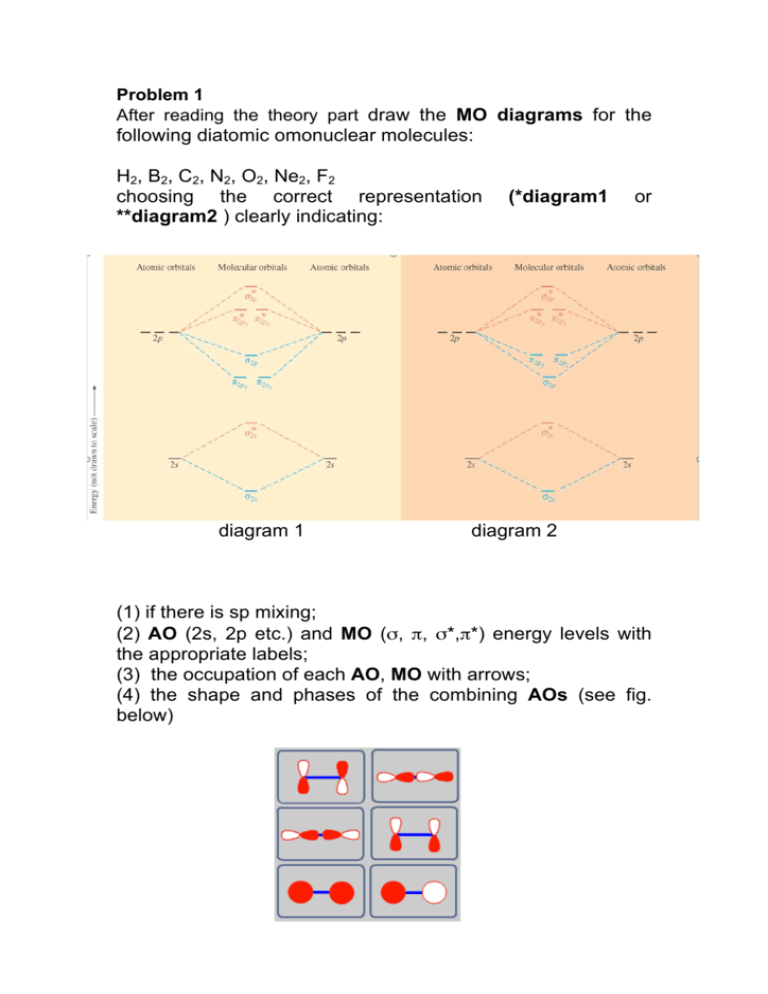
Problem 1 After reading the theory part draw the MO diagrams for the following diatomic omonuclear molecules: H2, B2, C2, N2, O2, Ne2, F2 choosing the correct representation **diagram2 ) clearly indicating: diagram 1 (*diagram1 or diagram 2 (1) if there is sp mixing; (2) AO (2s, 2p etc.) and MO (σ, π, σ*,π*) energy levels with the appropriate labels; (3) the occupation of each AO, MO with arrows; (4) the shape and phases of the combining AOs (see fig. below) H2 MO Diagram B2 MO Diagram C2 MO Diagram N2 MO Diagram O2 MO Diagram Ne2 MO Diagram MO Diagram F2 (5) Complete the following table (calculate all the quantities by considering by neglecting the 1s shell). B2 C2 N2 O2 Ne2 F2 electron configuration N. OF OCCUPIED MO N. of bonding electrons1 N. of antibonding electrons2 spin multiplicity3 Bond Order4 1. Number of electrons in bonding orbitals. 2. Number of electrons in antibonding orbitals. 3. spin multiplicity = (2S+1) where S represents the spin quantum number. 4. [(number of electrons in bonding levels) - (number of electrons in antibonding levels)] / 2 Problem 2 Below is an MO diagram of a π bond using two p orbitals. A nodal plane is a plane of zero electron density formed when orbitals of opposite phases overlap. It is indicated on the diagram below by a dashed line. 1. How many individual AOs are shown in this diagram? 2. How many electrons are present in each atomic orbital? 3. How do the energies of the bonding and antibonding MOs compare? 4. Another difference between the bonding MO and the antibonding MO is the presence of a nodal plane. Which MO contains this node? 5. For this π bond, put the electrons in the appropriate molecular orbital(s). Explain why you chose this configuration. 6. From this diagram, does in-phase overlap (both the same color) or out-of-phase overlap (one of each color) have to be present for a bonding interaction? Problem 3 Download the file C2H4.out under lecture material. This file is containing all the informations needed to visualize the MOs of the molecule C2H4. In oder to do that, open this file with the program wxMacMolPlt under application: go to Subwindow >> Surfaces >> 3D Orbital set initially the Contour Value to 0.004 (you can modify this any moment) then select Orb. and press Update in the end. Visualize all occupied molecular orbitals of C2H4 plus the 1st unoccupied orbital (LUMO). How many occupied orbitals you have? Fill in the following table numbering the orbital in ascending order. # Orbital 1 2 3 4 5 6 7 8 9 Energy Bonding Antibonding Orbital Type (σ/π/σ∗/π∗) Problem 4 Download the file C2H2.out under lecture material. This file is containing all the informations needed to visualize the MOs of the molecule C2H2. In oder to do that, open this file with the program wxMacMolPlt under application: go to Subwindow >> Surfaces >> 3D Orbital set in initially the Contour Value to 0.004 (you can modify this any moment) then select Orb. and press Update in the end. Visualize all occupied molecular orbitals of C2H2 plus the 1st unoccupied orbital (LUMO). How many occupied orbitals you have? Fill in the following table numbering the orbital in ascending order. # Orbital 1 2 3 4 5 6 7 8 Energy Bonding Antibonding Orbital Type (σ/π) Problem 5 Download the file C2H6.out under lecture material. This file is containing all the informations needed to visualize the MOs of the molecule C2H6. In oder to do that, open this file with the program wxMacMolPlt under application: go to Subwindow >> Surfaces >> 3D Orbital set in initially the Contour Value to 0.004 (you can modify this any moment) then select Orb. and press Update in the end. Visualize all occupied molecular orbitals of C6H6 plus the 1st unoccupied orbital (LUMO). How many occupied orbitals you have? Fill in the following table numbering the orbital in ascending order. # Orbital Energy Bonding Antibonding Orbital Type (σ/π)