Average Atomic Mass Calculation: Chemistry Presentation
advertisement

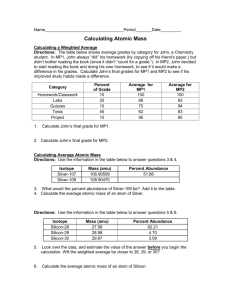
Average Atomic Mass http://tinyurl.com/md228mj Due Monday Oct 14, 2013 Objective • Calculate the average atomic mass for an element Average Atomic Mass The weighted average mass of all the isotopes found in nature for that element. Average Atomic Mass • Measured in atomic mass units (amu) Mass Spectrometer Instrument that determines the percentages and individual masses of each isotope Calculation The average atomic mass is calculated by the percent of each isotope’s mass [(mass of isotope) (%abundance)] + [(mass of isotope) (%abundance)] + [(mass of isotope) (%abundance)] … Practice Problem 1 Boron has two isotopes: Boron-10 and Boron-11. In nature 19% of Boron is Boron10, and 81% is Boron-11. Practice Problem 2 A new element, Tyserium (Ty), has recently been discovered on Mars and consists of two isotopes. One isotope has a mass of 331 amu and is 35.0 % abundant. The other isotope is 339 amu and is 65.0 % abundant. What is the mass of Ty as it appears on the periodic table? Logic Problem #1 Copper has an average atomic mass of 63.546 amu and only exists as Copper63 and Copper-65. What is the isotopic mass of the most common isotope of Copper? Logic Problem #2 The relative atomic mass of chlorine is 35.5 amu. What does this tell you about the relative abundance of the two naturally occurring isotopes of chlorine, Chlorine-35 and Chlorine-37. Check Yourself! • Make sure your calculated average atomic mass is within the range of the given mass numbers – X-230, X-235, X-240 • Rationalize your results – 90% of X-230, 5% of X-235, 5% of X-240 • Check your answers on the periodic table Questions? If you have any questions, jot them down!