Isotope (type of bolt)
advertisement

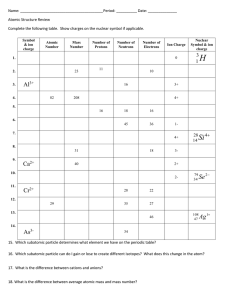
Lab 8 The Atomic Mass of “Boltium” Date: Objective Calculate the atomic mass of “Boltium” Background The atomic mass of any element is the average of the masses of all the naturally occurring isotopes of an element, weighted according to the abundance of each isotope. Take copper for example… Take any sample of pure copper and run it through a mass spectrometer… No matter where the copper came from, it will separate into the following isotopes: 69.17%: Copper-63, 62.939598 amu 30.83%: Copper-65, 64.927793 amu The atomic mass is calculated as a weighted average… 62.939598 x 0.6917 + 64.927793 x 0.3083 = 63.55 (rounded) Experiment Boltium sample: 50 atoms, separate into 4 different isotopes (types of bolts) Calculate in Analysis section Isotope Isotope Count Total Mass (g) Average Mass (g) % Abundance (type of bolt) (number of bolts of each type) (mass of pile) (Analysis #1) (Analysis #2) Analysis 1. Calculate the average mass of each isotope, record this in the data table. 2. Calculate the percent abundance of each isotope, record this in the data table. 3. Calculate the atomic mass of “Boltium.” This is a weighted average that factors in the abundance and mass of each isotopes of the element. Results Questions 1. Explain why the atomic mass of an atom should be a weighted average. 2. The atomic mass of carbon is 12.011 amu, although no atom of carbon actually weighs this amount. Explain why this is so. 3. What did each bolt in this lab represent? 4. This activity was a simulation of how isotopes affect the average atomic masses calculation. Describe 2 flaws of this simulation.