Cu 2+ + 2e - Solon City Schools
advertisement

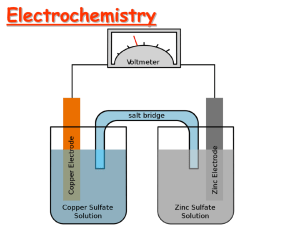
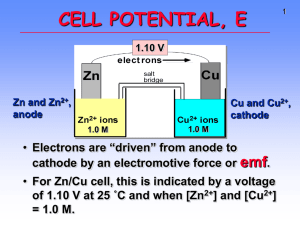
Electrochemistry Terminology Oxidation – A process in which an element attains a more positive oxidation state Na(s) Na+ + eReduction – A process in which an element attains a more negative oxidation state Cl2 + 2e- 2ClOxidizing agent -The substance that is reduced is the oxidizing agent Reducing agent - The substance that is oxidized is the reducing agent LEO says GER Electrochemistry Terminology Anode -The electrode where oxidation occurs Cathode - The electrode where reduction occurs Leo is a Reduction at the Cathode Step 1: Step 2: Step 3: 2ee- Step 4: Multiply each half-reaction by a factor so that the reducing agent supplies as many electrons as the oxidizing agent requires. Reducing agent Cu ---> Cu2+ + 2eOxidizing agent 2 Ag+ + 2 e- ---> 2 Ag Step 5: Add half-reactions to give the overall equation. Cu + 2 Ag+ ---> Cu2+ + 2Ag The equation is now balanced for both charge and mass. Balance the following in acid solution— VO2+ + Zn ---> VO2+ + Zn2+ Step 1: Write the half-reactions Ox Zn ---> Zn2+ Red VO2+ ---> VO2+ Step 2: Balance each half-reaction for mass. Ox Zn ---> Zn2+ Red 2 H+ + VO2+ ---> VO2+ + H2O Add H2O on O-deficient side and add H+ on other side for H-balance. Step 3: Balance half-reactions for charge. Ox Zn ---> Zn2+ + 2eRed e- + 2 H+ + VO2+ --> VO2+ + H2O Step 4: Multiply by an appropriate factor. Ox Zn ---> Zn2+ + 2eRed 2e- + 4 H+ + 2 VO2+ ---> 2 VO2+ + 2 H2O Step 5: Add balanced half-reactions Zn + 4 H+ + 2 VO2+ ---> Zn2+ + 2 VO2+ + 2 H2O Draw it! A great example of a thermodynamically spontaneous reaction is the thermite reaction. Here, iron oxide (Fe2O3 = rust) and aluminum metal powder undergo a redox (reduction-oxidation) reaction to form iron metal and aluminum oxide (Al2O3 = alumina): Fe2O3(s) Fe = +3 + Al(s) ↔ Al2O3(s) Al = 0 Al = +3 + Fe(l) Fe = 0 1) 2) 3) 4) 5) 12 5 2 30 10 1. 2. 3. 4. 5. Zn → Zn2+ + 2e– Zn2+ + 2e– → Zn Zn2+ + Cu → Zn + Cu2+ Zn + Cu2+ → Zn2+ + Cu two of these 1. 2. 3. 4. 5. S(s) NO3–(ag) Cr2O72–(aq) I– (aq) MnO4– (aq) An apparatus that allows a redox reaction to occur by transferring electrons through an external connector. Product favored reaction --> voltaic or galvanic cell ---> electric current Reactant favored reaction --> electrolytic cell ---> electric current used to cause chemical change. Batteries are voltaic cells Basic Concepts of Electrochemical Cells Anode Cathode potential Eo +1.10 V STANDARD CELL POTENTIAL, Zn(s) ---> Zn2+(aq) + 2eCu2+(aq) + 2e- ---> Cu(s) -------------------------------------------Cu2+(aq) + Zn(s) ---> Zn2+(aq) + Cu(s) Measuring Standard Electrode Potential Potentials are measured against a hydrogen ion reduction reaction, which is arbitrarily assigned a potential of zero volts. Table of Reduction Potentials Measured against the Standard Hydrogen Electrode oxidizing ability of ion Eo (V) Cu2+ + 2e- Cu +0.34 2 H+ + 2e- H2 0.00 Zn2+ + 2e- Zn -0.76 To determine an oxidation from a reduction table, just take the opposite sign of the reduction! reducing ability of element + Anode, negative, source of electrons Cathode, positive, sink for electrons Zn(s) ---> Zn2+(aq) + 2eEo = +0.76 V Cu2+(aq) + 2e- ---> Cu(s) Eo = +0.34 V --------------------------------------------------------------Cu2+(aq) + Zn(s) ---> Zn2+(aq) + Cu(s) Eo = +1.10 V All ingredients are present. Which way does reaction proceed? e- All ingredients are present. Which way does reaction proceed? Assume I- ion can reduce water. 2 H2O + 2e- ---> H2 + 2 OH2 I- ---> I2 + 2e------------------------------------------------2 I- + 2 H2O --> I2 + 2 OH- + H2 Cathode Anode -1.363 V Negative E˚ means rxn. occurs in opposite direction (the connection is backwards or you are recharging the battery) Galvanic (Electrochemical) Cells Spontaneous redox processes have: A positive cell potential, E0 A negative free energy change, (-G) Zn - Cu Galvanic Cell From a table of reduction potentials: Zn2+ + 2e- Zn Cu2+ + 2e- Cu E = -0.76V E = +0.34V Zn - Cu Galvanic Cell The less positive, or more negative reduction potential becomes the oxidation… Zn Zn2+ + 2eCu2+ + 2e- Cu Zn + Cu2+ Zn2+ + Cu E = +0.76V E = +0.34V E0 = + 1.10 V Line Notation An abbreviated representation of an electrochemical cell Zn(s) | Zn2+(aq) (1.0M) Anode Anode | material solution || Cu2+(aq) || Cathode solution Line notation is cool, just like AC (1.0M) | | Cu(s) Cathode material Zn(s) | Zn2+(aq) (1.0M) || H+(aq) (1.0M) |H2(g) (1.00 atm) | Pt(s) Calculating G0 for a Cell G0 = -nFE0 n = moles of electrons in balanced redox equation F = Faraday constant = 96,485 coulombs/mol e- Zn + Cu2+ Zn2+ + Cu E0 = + 1.10 V coulombs Joules G ( 2 mol e )(96 485 )(1.10 ) mol e Coulomb 0 G 212267 Joules 212 kJ 0 Day 3 (dahditdadahditahh…Charge!) The Nernst Equation Standard potentials assume a concentration of 1.0 M. The Nernst equation allows us to calculate potential when the two cells are not 1.0 M. RT EE ln( Q) nF R = 8.31 J/(molK) 0 T = Temperature in K n = moles of electrons in balanced redox equation F = Faraday constant = 96,485 coulombs/mol e- Nernst Equation Simplified At 25 C (298 K) the Nernst Equation is simplified this way: 0.0591 EE log( Q) n 0 Try Problem 62 Equilibrium Constants and Cell Potential At equilibrium, forward and reverse reactions occur at equal rates, therefore: 1. The battery is “dead” 2. The cell potential, E, is zero volts Modifying the Nernst Equation (at 25 C): 0.0591 0 volts E log( K ) n 0 Calculating an Equilibrium Constant from a Cell Potential Zn + Cu2+ Zn2+ + Cu E0 = + 1.10 V 0.0591 0 volts 1.10 log( K ) 2 (1.10)( 2) log( K ) 0.0591 37.2 log( K ) 10 37.2 K 1.58 x10 37 Let’s Do Problems: 26a, 28a, 32a, 38a Apply to Problem 70 ??? Concentration Cell Both sides have the same components but at different concentrations. Step 1: Determine which side undergoes oxidation, and which side undergoes reduction. ??? Anode Concentration Cell Cathode Both sides have the same components but at different concentrations. The 1.0 M Zn2+ must decrease in concentration, and the 0.10 M Zn2+ must increase in concentration Zn2+ (1.0M) + 2e- Zn (reduction) Zn Zn2+ (0.10M) + 2eZn2+ (1.0M) Zn2+ (0.10M) (oxidation) ??? Concentration Cell Anode Cathode Concentration Cell Both sides have the same components but at different concentrations. Step 2: Calculate cell potential using the Nernst Equation (assuming 25 C). Zn2+ (1.0M) Zn2+ (0.10M) 0.0591 EE log(Q) n 0 Nernst Calculations Zn2+ (1.0M) Zn2+ (0.10M) 0.0591 EE log( Q) n 0 E 0.0 Volts n 2 0 (0.10) Q (1.0) 0.0591 0.10 E 0.0 log( ) 0.030 Volts 2 1.0 Nearly same reactions as in common dry cell, but under basic conditions. Anode (-): Zn + 2 OH- ---> ZnO + H2O + 2eCathode (+): 2 MnO2 + H2O + 2e- ---> Mn2O3 + 2 OH- Anode: Cathode: Anode (-) Cathode (+) Anode (-) Cd + 2 OH- ---> Cd(OH)2 + 2eCathode (+) NiO(OH) + H2O + e- ---> Ni(OH)2 + OH- The positive electrode is made of Lithium cobalt oxide, or LiCoO2 The negative electrode is made of carbon. The Electrochemical Corrosion of Iro Electrolytic Processes Electrolytic processes are NOT spontaneous. They have: A negative cell potential, (-E0) A positive free energy change, (+G) Electrolysis of Water In acidic solution Anode rxn: 2 H 2O O2 4 H 4e Cathode rxn: 4 H 2O 4e 2 H 2 4OH 2 H 2O 2 H 2 O2 -1.23 V -0.83 V -2.06 V Electroplating of Silver Anode reaction: Ag Ag+ + eCathode reaction: Ag+ + e- Ag Electroplating requirements: 1. Solution of the plating metal 2. Anode made of the plating metal 3. Cathode with the object to be plated 4. Source of current Calculating plating Step 1 – convert current and time to quantity of charge in coulombs a. Amps x time = total charge transferred in coulombs (Coulomb/sec) x sec = coulombs Step 2 – convert quantity of charge in coulombs to moles of electrons coulombs /(96,485 coulombs/mol e-) = mol eStep 3 – Convert moles of electrons to moles of substance mol e- x (mole substance/mol e-) = mol substance Step 4 – Convert moles of substance to grams of substance mol substance x formula mass of substance = mass of substance Suppose that in starting a car on a cold morning a current of 125 amperes is drawn for 15.0 seconds from a cell of the type described above. How many grams of Pb would be consumed? (The atomic weight of Pb is 207.19.) 2+ Pb 125 C 15 sec 1 sec + 2e Pb 1 mol e- 1 mol Pb 207.19 g - 1 mol Pb 2 mol e 96 485 C 2.01 g Pb Solving an Electroplating Problem Q: How many seconds will it take to plate out 5.0 grams of silver from a solution of AgNO3 using a 20.0 Ampere current? Ag+ + e- Ag 5.0 g 1 mol Ag 1 mol e- 96 485 C 1 s 20.0 C 1 mol e 107.87 g 1 mol Ag = 2.2 x 102 s Do 79!!! Do 81!!!