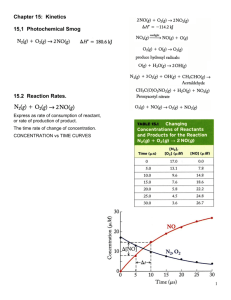
Chemical Kinetics
advertisement

Chemical Kinetics The area of chemistry that concerns reaction rates. 1 Collision Model Key Idea: Molecules must collide to react and must be oriented the correct way. However, only a small fraction of collisions produces a reaction. Why? Arrhenius: An activation energy must be overcome. 2 Reaction Rate Change in concentration (conc) of a reactant or product per unit time. conc of A at time t2 conc of A at time t1 Rate = t2 t1 A Why does this formula t look so familiar? 3 For example 2NO2 2NO + O2 As NO2 is used up more NO and O2 should appear. How much O2 appears when 3.0 mol of NO2 disappears? 4 5 6 Factors affecting rate of rxn Temperature- for every 10°C increase, the rate of the reaction will double (approximation) [Reactants] – allows for more collisions. Catalyst – speeds up by lowering activation energy. Nature of reactants – Surface Area mainly but also complexity of the molecule. 7 Rate Laws Rate = k[NO2]n k = rate constant n = rate order – affect of changing the [Reactant] on [Product] k and n can only be found experimentally. 8 Method of Initial Rates Initial Rate: the “instantaneous rate” just after the reaction begins. This is a cheat so we don’t have to worry about the reverse rxn. The initial rate is determined in several experiments using different initial concentrations. 9 You can figure out order of each reactant and then figure out value of k. 10 Overall Reaction Order Sum of the order of each component in the rate law. rate = k[H2SeO3][H+]2[I]3 The overall reaction order is 1 + 2 + 3 = 6. 11 Types of Rate Laws Differential Rate Law: expresses how rate depends on concentration. What we call the rate law. Integrated Rate Law: expresses how concentration depends on time. Lets you figure out concentrations of stuff after a certain amount of time. 12 First-Order Rate Law Find it in the equation sheet For A Products in a 1st-order reaction, A Rate = k A t Integrated first-order rate law is ln[A]t = kt + ln[A]o Notice this is slope intercept formula 13 Half-Life of a First-Order Reaction 0.693 t1/2 k t1/2 = half-life of the reaction Half life of a reaction is the time required for a reactant to reach half its original concentration k = rate constant For a first-order reaction, the half-life does not depend on concentration. (This is important) 14 Second-Order Rate Law For A products in a second-order reaction, A Rate = k A 2 t Integrated rate law is (slope intercept again) 1 1 kt + A Ao 15 Half-Life of a Second-Order Reaction t1/2 1 kA o t1/2 = half-life of the reaction k = rate constant Ao = initial concentration of A The half-life is dependent upon the initial concentration and changes over time 16 Zero order rxns 1. Rate is constant; it does not change with changing concentration 2. Zero order sometimes happens with catalysis 3. The integrate rate law is NOT found on the formula sheet. 17 Reaction Mechanism The series of steps by which a chemical reaction occurs. A chemical equation does not tell us how reactants become products - it is a summary of the overall process. 18 Reaction Mechanism (continued) The reaction 6CO 2 6H 2 O light C 6 H12 O 6 6O 2 has many steps in the reaction mechanism. 19 Often Used Terms Intermediate: formed in one step and used up in a subsequent step and so is never seen as a product. Molecularity: the number of species that must collide to produce the reaction indicated by that step. Elementary Step: A reaction whose rate law can be written from its molecularity. uni, bi and termolecular 20 Elementary step example 2 NO2 + F2 2 NO2F follows the mechanism, NO2 + F2 NO2F + F NO2 + F NO2F 21 Rate-Determining Step In a multistep reaction, it is the slowest step. It therefore determines the rate of reaction. 22 Writing a rate law from an elementary step 2 NO2 + F2 2 NO2F follows the mechanism, NO2 + F2 NO2F + F slow NO2 + F NO2F fast The rate law can be written????? 23 Tricks If it says fast equilibrium then you substitute Overall reaction 2NO + O2 2NO2 Step 1 2 NO N2O2 (fast equilibrium) Step 2 N2O2 + O2 2NO2 slow Rate law = k[NO]2 [O2] 24 Tricks Part Dos Remember all the elementary steps must add up to the chemical reaction. 25 Molecularity Elementary step Rate law 1 A -> products rate = k [A] 2 A + A -> products rate = k [A]2 2 A + B -> products rate = k [A] [B] 3 A + A + A -> products rate = k [A]3 3 A + 2 B -> products rate = k [A] [B]2 3 A + B + C -> products rate = k [A] [B] [C] 26 Arrhenius Equation Collisions must have enough energy to produce the reaction (must equal or exceed the activation energy). 27 A graph showing two steps. Blue is uncatalyzed 28 Catalysis Catalyst: A substance that speeds up a reaction without being consumed. It does so by lowering the activation energy. Enzyme: A large molecule (usually a protein) that catalyzes biological reactions. 29 [NO2] what order? time (s) [] ln 1/[] 0 0.5 -0.69315 2 1200 0.444 -0.81193 2.252252 3000 0.381 -0.96496 2.624672 4500 0.34 -1.07881 2.941176 9000 0.25 -1.38629 4 18000 0.174 -1.7487 5.747126 30 [] 0.6 0.4 0.2 0 0 5000 10000 15000 20000 1/[] 8 6 4 2 0 0 ln 5000 10000 15000 20000 0 0 5000 10000 15000 20000 -1 -2 31 Catalysts Homogeneous (same phase) vs Heterogenous (different phases) The difference between an intermediate and a catalsyt is? 32 Think about your areas of strength and weakness… 1. Figure out order from a set of data and write a rate law. 2. Calculate the value of k including units. 3. Use and understand integrated rate laws. 4. Determine rate law from elementary steps. 5. Know how catalysts work 6. Understand misc. vocabulary. 33



![1 [A]](http://s3.studylib.net/store/data/009563931_1-7a369d18638f75aaafb200fb61a0b929-300x300.png)



