Collision Theory, Elementary Steps, Intermediates, Rate Determining Steps
advertisement

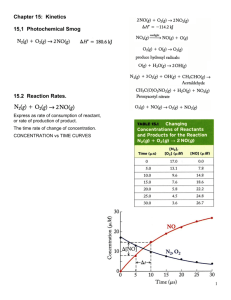
Collision Theory, Elementary Steps, Intermediates, Rate Determining Steps Page 387 # 1 390 # 2 391 # 1 - 4 Prepare Iodine Clock Lab RATE DETERMINING STEP Many reactions consist of a series of separate stages. Each step has its own rate and rate constant. The overall rate of a multi-step process is governed by the slowest step (like a production line where overall output can be held up by a slow worker). This step is known as the RATE DETERMINING STEP . If there is more than one step, the rate equation may not contai n all the reactants in its format. RATE DETERMINING STEP THE REACTION BETWEEN PROPANONE AND IODINE Iodine and propanone react in the presence of acid CH 3 COCH 3 + I 2 The rate equation is... r = k [CH 3 COCH 3 ] [H + ] Why do H + ions appear in the rate equation? Why does I 2 not appear in the rate equation? CH 3 COCH 2 I + HI RATE DETERMINING STEP THE REACTION BETWEEN PROPANONE AND IODINE Iodine and propanone react in the presence of acid CH 3 COCH 3 + I 2 CH 3 COCH 2 I + HI The rate equation is... r = k [CH 3 COCH 3 ] [H + ] Why do H + ions appear in the rate equation? The reaction is catalysed by acid [H + ] affects the rate but is unchanged overall Why does I 2 not appear in the rate equation? The rate determining step doesn't involve I 2 RATE DETERMINING STEP THE REACTION BETWEEN PROPANONE AND IODINE Iodine and propanone react in the presence of acid CH 3 COCH 3 + I 2 CH 3 COCH 2 I + HI The rate equation is... r = k [CH 3 COCH 3 ] [H + ] Why do H + ions appear in the rate equation? The reaction is catalysed by acid [H + ] affects the rate but is unchanged overall Why does I 2 not appear in the rate equation? The rate determining step doesn't involve I 2 The slowest step of any multi-step reaction is known as the rate determining step and it is the species involved in this step that are found in the overa ll rate equation. Catalysts appear in the rate equation because they affect the ra te but they do not appear in the stoichiometric equation because they remain chemically unchanged RATE DETERMINING STEP HYDROLYSIS OF HALOALKANES OH - Haloalkanes (general formula RX) are hydrolysed by hydroxide ion to give alcohols. RX + With many haloalkanes the rate equation is... This is because both the RX and OH - must collide for a reaction to take place in ONE STEP r = k [RX][OH -] ROH + X- SECOND ORDER RATE DETERMINING STEP HYDROLYSIS OF HALOALKANES OH - RX + With many haloalkanes the rate equation is... This is because both the RX and OH - must collide for a reaction to take place in ONE STEP r = k [RX][OH -] SECOND ORDER but with others it only depends on [RX]... r = k [RX] FIRST ORDER The reaction has taken place in TWO STEPS... - the first involves breaking an R-X bond - the second step involves the two ions joining i) ii) ROH + X- Haloalkanes (general formula RX) are hydrolysed by hydroxide ion to give alcohols. RX R+ + XR + + OH ROH Slow Fast The first step is slower as it involves bond breaking and energy has to be put in. The first order mechanism is favoured by tertiary haloalkanes because the hydroxide ion is hindered in its approach by alkyl groups if the mechanism involves the hy droxide ion and haloalkane colliding. RATE DETERMINING STEP The reaction H 2 O 2 + 2H 3 O + 2 I+ Step 1 H 2 O 2 +I Step 2 IO Step 3 HIO + H 3 O + I+ +H 3 O + I2 + 4H 2 O IO HIO takes place in 3 steps +H 2 O SLOW + FAST H 2O I2 + 2H 2 O The rate determining step is STEP 1 as it is the slowest FAST RATE DETERMINING STEP The reaction H 2 O 2 + 2H 3 O + 2 I+ I2 + 4H 2 O Step 1 H 2 O 2 +I IO Step 2 IO Step 3 HIO + H 3 O + I+ +H 3 O + HIO takes place in 3 steps +H 2 O SLOW + FAST H 2O I2 + 2H 2 O FAST The rate determining step is STEP 1 as it is the slowest The reaction 2N 2 O 5 4NO 2 + O 2 Step 1 N2O 5 Step 2 NO 2 + NO 3 Step 3 NO + takes place in 3 steps NO 2 + NO 3 NO 3 SLOW NO + NO 2 + O 2 2NO 2 The rate determining step is STEP 1 from another Step 1 FAST FAST rate = k [N2O5] OTHER TOPICS Autocatalysis A small number of reactions appear to speed up, rather than slow down, for a time. This is because one of the products is acting as a catalyst and as more product is formed the reaction gets faster. One of the best known examples is the catalytic properties of Mn 2+ (aq) on the decomposition of MnO 4 (aq). You will notice it in a titration of KMnO 4 with either hydrogen peroxide or ethanedioic (oxalic) acid. Molecularity e.g. The number of individual particles of the reacting species takin g part in the rate determining step of a reaction A + 2B A C+D 2B molecularity is 3 - one A and two B's need to collide however has a molecularity of 1 - only one A is involved Rate Laws and Elementary Steps Unimolecular reaction A products rate = k [A] Bimolecular reaction A+B products rate = k [A][B] Bimolecular reaction A+A products rate = k [A]2 Reaction Mechanisms The overall progress of a chemical reaction can be represented at the molecular level by a series of simple elementary steps or elementary reactions. The sequence of elementary steps that leads to product formation is the reaction mechanism. 2NO (g) + O2 (g) 2NO2 (g) N2O2 is detected during the reaction! Elementary step: NO + NO N2O2 + Elementary step: N2O2 + O2 2NO2 Overall reaction: 2NO + O2 2NO2 Reaction Intermediates Intermediates are species that appear in a reaction mechanism but not in the overall balanced equation. An intermediate is always formed in an early elementary step and consumed in a later elementary step. Elementary step: NO + NO N2O2 + Elementary step: N2O2 + O2 2NO2 Overall reaction: 2NO + O2 2NO2 Rate Laws and Rate Determining Steps Writing plausible reaction mechanisms: • The sum of the elementary steps must give the overall balanced equation for the reaction. • The rate-determining step should predict the same rate law that is determined experimentally. The experimental rate law for the reaction between NO 2 and CO to produce NO and CO2 is rate = k[NO2]2. The reaction is believed to occur via two steps: Step 1: NO 2 + NO2 NO + NO3 Step 2: NO3 + CO NO2 + CO2 What is the equation for the overall reaction? NO2+ CO NO + CO2 What is the intermediate? Catalyst? NO3 NO2 What can you say about the relative rates of steps 1 and 2? rate = k[NO2]2 is the rate law for step 1 so step 1 must be slower than step 2 Write the rate law for this reaction. Rate = k [HBr] [O2] List all intermediates in this reaction. HOOBr, HOBr List all catalysts in this reaction. None Ostwald Process 4NH3 (g) + 5O2 (g) 2NO (g) + O2 2NO2 (g) + H2O (l) Pt catalyst 4NO (g) + 6H2O (g) 2NO2 (g) (g) HNO2 (aq) + HNO3 (aq) Pt-Rh catalysts used in Ostwald process Hot Pt wire over NH3 solution © 2003 JONATHAN HOPTON & KNOCKHARDY PUBLISHING