Chemical Reactions: Types, Equations, and Formulas
advertisement
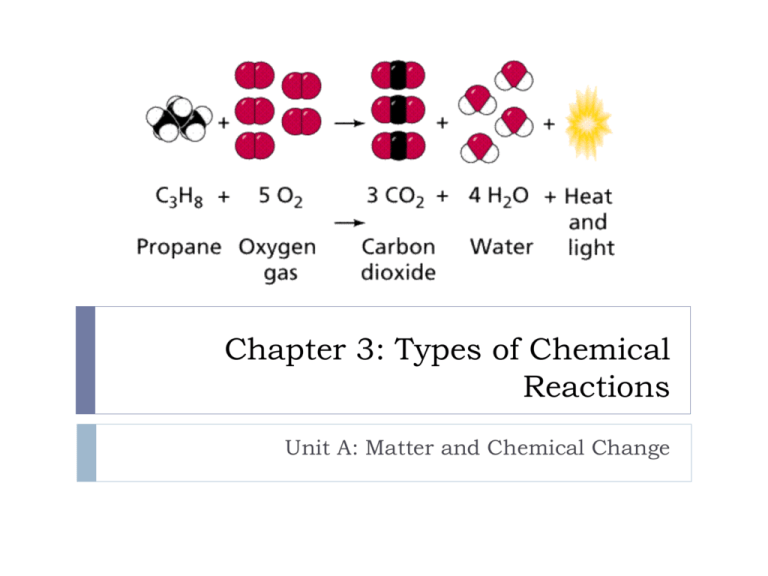
Chapter 3: Types of Chemical Reactions Unit A: Matter and Chemical Change What we already know… Chemical reactions can be simple or complex Chemical changes can be obvious or less noticeable Chemical reactions can occur immediately or more slowly To describe chemical reactions, we use word equations 3.1 Word Equations Word equations are always written in the same format Left side of the equation lists the reactants Right side of the equation lists the products Arrow points from the reactants to the products All Reactants All Products All the reactants and all the products are separated by a plus sign (+) reactant 1 + reactant 2 product 1 + product 2 3.1 Word Equations Oxygen + food (glucose) Continued carbon dioxide + water + energy Carbonic acid and calcium carbonate produce calcium hydrogen carbonate 3.2 Types of Reactions In a simple composition reaction, two or more simple elements combine to form a compound. Element1 + Element 2 A + B magnesium + oxygen Compound AB magnesium oxide + light + thermal energy Most simple composition reactions are exothermic 3.2 Types of Reactions Continued In a simple decomposition reaction, a reaction breaks a compound into its component elements. Compound AB water + electric energy Element1 + Element 2 A + B hydrogen gas + oxygen gas Most decomposition reactions are endothermic 3.2 Types of Reactions Combustion reactions always occur in the presence of oxygen. Continued Candle wax burning in the presence of oxygen produces carbon dioxide and water Neutralization reactions are an acid reacting with a base to produce a form of salt and water. acid + base salt + water 3.3 Formulas for Common Compounds sulfur + zinc zinc sulfide A chemical name is the standard scientific name by which each element or compound is known; established by an internationally recognized naming system. A chemical formula is the standard scientific symbol by which each chemical element or compound is known. S8(s) + 8Zn(s) 8ZnS(s) 3.3 Formulas for Common Compounds Continued Many chemical compounds are often identified by a common name. Easier to use and have existed before the international naming system Ethanol - alcohol isopropyl alcohol – rubbing alcolhol Sodium bicarbonate – baking soda ethylene glycol - antifreeze Methane – natural gas sodium chloride - salt Dihydrogen oxide - water sucrose - sugar 3.4 Chemical Equations The Law of Conservation of Mass states: In a chemical reaction, matter is not created or destroyed. If you start a reaction with 10g of reactants, you will end up with 10g of products. ~ Antoine Lavoisier (1743-1794) vinegar + baking soda sodium acetate + water + carbon dioxide gas CH3COOH(aq) + NaHCO3(s) NaC2H3O2(l) + H2O(l) + CO2(g)











