File - Mrs. Coyle's College Chemistry
advertisement

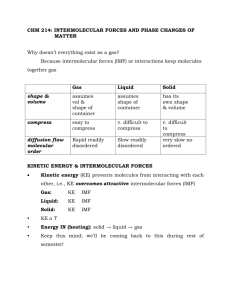
Properties of Liquids College Chemistry KINETIC MOLECULAR THEORY OF GASES Back when we studied gases, we used the KMT to explain the behavior of gases Distance between molecules are so great that at ordinary temperature and pressure, there is no interaction between molecules Lots of empty space gives gases lots of their properties Compressibility Expandability Low densities Kinetic-Molecular Theory Applied to Liquids and Solids Condensed states – liquids and solids, referred to as this because these states have much higher densities than they do in the gaseous state We have already looked at how the kinetic-molecular theory can be applied to gases, how can it be applied to liquids and solids? Kinetic-Molecular Theory Applied to Liquids and Solids Gas Liquid Solid Highly compressible Slightly compressible Very little compressibility Low density High density High density Fills container completely Does not expand to fill – has a definite volume Rigidly retains its volume Assumes shape of container Assumes shape of container Retains own shape Rapid diffusion Slow diffusion Extremely low diffusion High expansion on heating Low expansion on heating Low expansion on heating KINETIC-MOLECULAR THEORY APPLIED TO SOLIDS AND LIQUIDS Densities of solids are usually (except for water) higher than liquids and it is possible for multiple phases of matter to exist at the same time Phase – homogeneous part of the system in contact with other parts of the system but separated by a well-defined boundary Ex: a glass of water with ice in it FORCES AND BONDS BETWEEN GASES AND LIQUIDS Intramolecular forces– chemical bonds, hold atoms together WITHIN a molecule Both covalent and ionic bonds Intermolecular forces – exist BETWEEN molecules General name: van der Waals forces very weak, but very important for life INTERMOLECULAR VS. INTRAMOLECULAR FORCES Intramolecular forces – hold atoms together (ionic, covalent, or metallic bonds) Intermolecular forces – attractive forces that hold molecules together Generally much weaker than intramolecular forces, but define the properties of the liquids nonetheless We can define these intermolecular forces overall as van der Waals forces, named after the Dutch physicist Johannes van der Waals All the following intermolecular forces we will talk about are van der Waals forces, except for hydrogen bonding DIPOLE - Review In simplistic terms, the shift of electron density to one side of the compound (the most electronegative side) Polar covalent bonds usually exhibit dipole behavior fairly well Not ionic enough to pull all electrons towards them like in ionic Not covalent enough to fully share electrons http://www.grandinetti.org/Te aching/Chem121/Lectures/VS EPR/ ION-DIPOLE ATTRACTIVE FORCES An ion is attracted to a polar molecule An anion is attracted to the partial positive side A cation is attracted to the partial negative side This “splits up” the ionic compound soluble This force becomes stronger as either the ion has a larger charge or the polar molecule becomes stronger (dipole) http://www.chem.purdue.edu/gche lp/liquids/iondip.html ION-DIPOLE FORCE One example of an ion-dipole interaction is hydration (interaction of cations and/or anions in water) Ions with a larger charge interact stronger with water because they have a stronger dipole Ex: Na+ vs. Mg+2 DIPOLE-DIPOLE FORCES Occurs between polar covalent molecules with permanent dipoles Larger the dipole, the larger the force The dipole of the molecule forms an attractions with the dipole of the water molecule Opposites attract! some covalent molecules will still not form conducting solutions (too strong of bonds) Very weak http://www.chem.purdue.edu/gchelp/liquids/dipdip.html DISPERSION FORCES A normal atom is purely symmetrical and the net charge of the atom is zero A temporary dipole can form that causes a separation of the positive and negative charges of the atom This temporary dipole can force the neighboring atoms to become distorted and form induced dipoles A dipole created by the presence of a neighboring dipole Results in a weak dispersion force that can hold atoms together Ex: at high temperatures, dispersion forces hold noble gases together and become condensed to liquids DISPERSION FORCES • Dispersion forces are THE only force between nonpolar molecules • Dispersion forces happen between ALL MOLECULES! • WEAKEST type of force http://www.chem.purdue.edu/gchelp/liquids/disperse.html DISPERSION FORCES At low enough temperatures (and reduced atom speeds), these dispersion forces are enough to hold atomic gases together causing the gas to condense DISPERSION FORCES Strength of dispersion forces depends on the charge of the ion (or the strength of the dipole) just like ion-dipole interactions However, polarizability of the molecule also plays a huge role Polarizability – ease with which the electron distribution in the atom can be distorted (think of a balloon) Usually the larger the # of electrons and the more diffuse the electron cloud is, the more polarizable it is (larger molar mass) DISPERSION FORCES Dispersion (or London) forces usually increase with molar mass b/c molecules with larger molar masses tend to have more electrons Dispersion forces increase in strength with the number of electrons Also, electrons are more easily disturbed b/c outer electrons aren’t held as strongly Usually the larger the molar mass (stronger the dispersion forces), the greater the melting/boiling point DISPERSION FORCES Melting Points of Similar Nonpolar Compounds Compound Melting Point (°C) CH4 -182.5 CF4 -150.0 CCl4 -23.0 CBr4 90.0 CI4 171.0 LONDON DISPERSION FORCES EXAMPLE 11.1 What types of intermolecular forces exist between the following pairs: (a) HBr and H2S, (b) Cl2 and CBr4 (a) HBr and H2S All molecules have dispersion forces Ion-dipole? No Dipole-dipole? Yes, it is polar (b) Cl2 and CBr4 All molecules have dispersion Ion-dipole? No Dipole-dipole? No, not polar HYDROGEN BONDING Hydrogen bonding occurs when an extremely strong dipole-dipole force occurs between H and either F, O, or N Must occur in very electronegative atoms! Why? Separation of dipoles It has a misleading name – NOT a bond, only an intermolecular force Hydrogen bonding is why water has a very high boiling point compared to other compounds Strongest type of bond! DIPOLE RELATED FORCES Between Ion – Dipole Ion charge and polar molecule H - bonding Polar bonded hydrogen and dipole charge Dipole - Dipole Ion – induced dipole Dipole – induced dipole Induced dipole – induced dipole (dispersion) Decreasing Strength Type Two polar molecules Ion charge and nonpolar molecule Polar and nonpolar molecule 2 nonpolar molecules Remember, ALL molecules have this! Stronger than dipole-related forces: Ionic, metallic, and covalent bonding Weaker than dipole-related forces: London dispersion forces Can mix and match terms! VISCOSITY Viscosity – “friction” or resistance to motion that exists between molecules of a liquid when they move past one another Stronger the attraction between molecules (intermolecular forces) = stronger resistance (greater viscosity) Viscosity increases as temperature increases This is why people heat up maple syrup – so it flows quicker Ex: alcohol and gasoline both “run” a lot faster than water, and much faster than syrup Alcohol and gasoline both have weaker intermolecular forces SURFACE TENSION Surface tension – imbalance of intermolecular forces at the surface of a liquid; these uneven forces make the surface behave as if it had a tight film stretched across it Surface tension explains the “beading” of raindrops, the ability of certain insects to walk across the water, etc. Surface tension increases in liquids with stronger intermolecular forces water has a VERY strong intermolecular forces SURFACE TENSION One example of surface tension is capillary action Water rising spontaneously up a tube (or water traveling up your jeans after you step in a puddle) Two types of forces bring about capillary action 1. cohesion 2. adhesion SURFACE TENSION Cohesion – intermolecular attraction between like molecules Think “co” – “cooperate” Example, water molecules Adhesion – attraction between unlike molecules If we think of water in a tube, this would be the attraction between water and the glass tube If adhesion is stronger than cohesion, the liquid will be pulled up Mercury however, will not do this STRUCTURE OF WATER Most interesting aspect: liquid water has a higher density than ice This is why ice floats Liquid water can form two hydrogen bonds, with each oxygen approximately tetrahedral bonded to four hydrogen (two covalent and two hydrogen) Ice cannot hydrogen bond because it has a highly ordered 3D structure and molecules can’t get to close to one another STRUCTURE OF WATER But let’s look at ice as it melts… At its melting point, the water has enough KE to pull free of intermolecular hydrogen bonds These molecules become trapped in the space of the 3D ice structure As a result, there is more molecules per unit volume in liquid water than ice At the same time, thermal expansion takes place (water expands as its been heating), so the density decreases Therefore, max density of water is actually at 4°C WHY DO LAKES FREEZE TOP DOWN? As we just talked about, as temperature of the water drops near the surface, density increases The colder water than sinks to the bottom and the less dense, warmer water rises to the top This continues until the overall water temperature is 4°C Below this temperature, density of water begins to slowly decrease with decreasing temperature and it no longer sinks Ice will form, but since it is less dense than water, it will not sink