Chemistry Homework Solutions: Intermolecular Forces & Solubility
advertisement

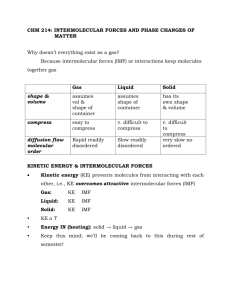
CHEMISTRY 161 HW CH#10A 24, 26, 28, 30, 36, 50, 52, 60, 66, 76, 80, 82, 86. 10-24 ICl is a polar molecule that has stronger intermolecular forces than the nonpolar molecules of Br 2, which have only the weak dispersion forces. It takes less energy to overcome weak intermolecular forces, so Br2 has a lower boiling point than ICl. 10-26 NH3 has the potential for hydrogen bonding due to the presence of one of the three very electronegative elements (F, O, and, in this case, N) whereas PH 3 does not. The higher boiling point of NH3 is due to the strong dipole–dipole interaction of the hydrogen bond. 10-28 From the Lewis structures of these molecules, we know that CO 2 is linear whereas all the other molecules are bent. CO2, therefore, is nonpolar and has only dispersion forces, the weakest intermolecular forces between its molecules. 10-30 If more atoms are present, more electron clouds around the atoms can be temporarily distorted to set up an attraction between molecules because of dispersion forces. The individual dispersion forces in a large molecule are probably about the same in strength as those of a smaller molecule. In a large molecule, however, there are many more of them. 10-36 (a) CO2 is a linear molecule and as such is nonpolar. For sublimation of CO2, only the weak dispersion forces are broken between the molecules. (b) CHCl3 is a polar, tetrahedral molecule. In order for this liquid to boil, dipole–dipole interactions between the molecules must be broken. Dispersion forces are also present, but they are weaker than the dipole–dipole interactions. (c) H2O is a polar, bent molecule with H bonded to the very electronegative O atom. In order to melt, hydrogen bonds (a special class of dipole–dipole interaction) between the water molecules must break. Dispersion forces are also present, but they are weaker than the hydrogen bonds. 10-50 (a) Br2 is nonpolar and NaBr is an ionic solid. Br2 is more soluble in CCl4. (b) C2H5OH is more polar (because of its OH group) than CH 3OCH3. CH3OCH3 is more soluble in CCl4. (c) CS2 is nonpolar and KOH is an ionic solid. CS2 is more soluble in CCl4. (d) I2 is nonpolar and CaF2 is an ionic solid. I2 is more soluble in CCl4. 10-52 We are given that Cgas = 6.5 mg/L. The solubility of O2 expressed in moles per liter is 1 mol O2 1g 6.5 mg/L 2.0 10–4 mol/L 1000 mg 32 g But the partial pressure of O2 in the atmosphere is 0.209 atm. Using this pressure in Henry’s law we find kH to be 2.0 10 –4 mol/L kH 9.6 10 –4 mol/L atm 0.209 atm 10-60 All of the molecules have nonpolar carbon–hydrogen chains, so we need to focus on the water interaction with the C—NH2 or C—X (X = halide). (a) CH3(CH2)2CH2NH2 has H bonded to electronegative N, which means it can form hydrogen bonds with H2O. These are generally stronger than dipole–dipole interactions. (b) CH3(CH2)4CH2Cl has a C—Cl polar bond, so the strongest interactions with water are dipole– dipole interactions. (c) CH3(CH2)6CH2Br has a C—Br polar bond, so the strongest interactions with water are dipole– dipole interactions. (d) CH3(CH2)8CH2I has a C—I bond, which is not polar, so the strongest interactions with water are dispersion forces. Because of its strong hydrogen bond interactions with water, CH3(CH2)2CH2NH2 (a) is the most soluble of these species in water. 10-66 A gas is present generally at high temperatures but making it dense (like a liquid) would mean that the pressure should be high. We would expect supercritical fluids to be present, then, at high temperatures and high pressures. This is indicated in a phase diagram as temperatures and pressures above the critical point. 10-76 (a) 0.32 atm and 120˚C: gas (b) 300 atm and 400˚C: supercritical fluid (c) 1 atm and 0˚C: solid and liquid 10-80 The cold needle would sink if the water were hot enough to disrupt the hydrogen bonding between water molecules. 1082 The fact that mercury does not rise at all in the capillary tube must mean that its cohesive forces are much stronger than its adhesive forces. In fact, the adhesive forces may be repelling so as to cause the mercury level to be lower than the pool of mercury that the capillary tube sits in. 10-86 The stronger the intermolecular forces, the more difficult it is to separate molecules in a liquid. This leads to high surface tension and high viscosity when the intermolecular forces in a liquid are strong.