Metric Measurement Review
advertisement

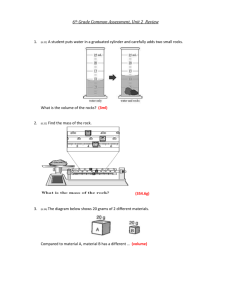
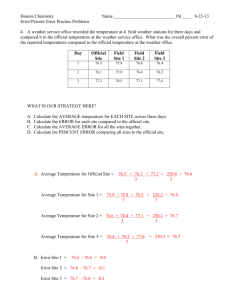
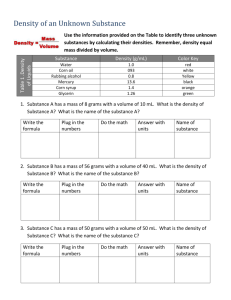
Approximately how many cells in the human body? 10 Trillion Approximately how many bacterium in a human body’s biome? 100 Trillion Metric Measurement Review Metric Prefixes: kilo- to mili- Metric Prefixes mL Liter mL are a small unit—how many drops per mL Liters are a larger unit Bigger unit of measurement= bigger or smaller number? Move decimal to the? 7000 mL Using Graduated Cylinders-Volume Value of graduation marks (lines) Distance between long lines Count lines & do simple math 40-20= 20 mL between long lines 10 lines between long lines 20/10= 2 mL per line Using Graduated Cylinders-Volume Value of graduation marks (lines) ? Reading a graduated cylinder Meniscus – The curve of the water Read at the BOTTOM of the curve Example=? Using Graduated Cylinders-Volume Take measurement as precise as equipment allows Estimate to the next place Example=? Volume of object= difference of first measurement and second Ex 70.5 & 75.0= difference/volume of ? Volume through dimensions & equation Volume= Length x Width x Height Thickness of slide 1mm = cm3 Human error Measurements Calculations More accurate when there are less steps required because there is less human error Always use equipment that allows you to get the answer you want if possible Graduated Cylinder for Volume instead of Ruler Volume by Formula Item too large to use graduated cylinder? Length x Width x Height Aquarium cm3=mL mLL Divide by 1,000 or move decimal to the left 3 places Lgallons Appropriate measurements Inside dimensions TRIPLE BEAM BALANCE • Zeroing balance BEFORE measurement • Pointer • Zero Mark • Adjustment Knob Triple Beam Balance LargestMediumSmall Add values of three arms to get total Example? Estimate one place further Comparing Balances Triple Beam Balance Electronic Balance Zero Zero More precise measurement Less precise 135.15 More human error 135.0 Less Human Error Precision of Measurement 100 grams vs. 100.0 grams 100 grams = 99.5-100.4 grams 100.0 grams= 100.0-100.04 Always measure as accurately as possible with equipment Estimate one place further Don’t give answers of around 2 when it is 2.33333 Percent Error The difference in the accepted value and your value Divide by accepted value Multiplied by 100 Your answer 10.5 10.5-10= .5 Accepted value 10 .5/10=.05 .05 x 100= 5.0% Color Challenge & Density Directions should have left you with rainbow with 10mL in each test tube Left over water in tubes Inaccurate measurements Mass & Volume Density Mass/Volume=Density g/cm3 Ruler Errors Element Flashcards Set 1 1. Hydrogen 2. Nitrogen 3. Neon 4. Carbon 5. Helium 6. Oxygen 7. Fluorine 8. Beryllium 9. Boron 10. Lithium O Turn Over Oxygen