Determine the Density of an unknown liquid
advertisement

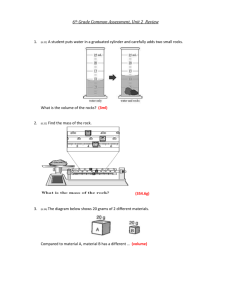
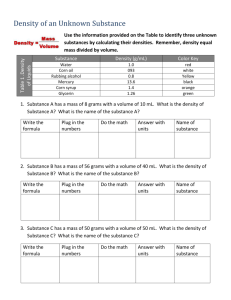
Determine the Density of an unknown liquid Name_____________________ Period_________ Unknown Number/Letter Hypothesis - Try to determine whether the density of your liquid is greater or less than 1 by placing a drop of the substance in a test tube filled with water. Complete the following table for the unknown substance. Data: Trial number mass of graduated cylinder plus liquid (grams) mass of graduated cylinder (grams) mass of liquid(grams) volume of liquid in (ml) Density of liquid (g/ml) 1 2 3 4 5 Average Density Use the list of densities provided on the back to compare the density of the unknown to the actual densities of several liquids. Use this list to identify the unknown Identity of unknown 1. Distilled Water 1.00 g/mL 2. Ethanol 0.81 g/mL 3. Isopropyl Alcohol 0.7825 g/mL 4. Vinegar 1.015 g/mL 5. Salt Water 1.177 g/mL 6. Sierra Mist 1.07 g/mL 7. Bleach 1.10 g/mL 8. Hydrogen Peroxide 1.44 g/mL 9. Methanol 0.7918 g/mL USE DIMENSIONAL ANALYSIS AND CORRECT SIG FIGS 1. A block of metal has a mass of 40 grams. Its volume is 10cm 3. What is its density? 2. Pure water has a density of 1.0 g/ml. What would be the mass of 50 ml of water? 3. a. Aluminum has a density of 2.7 g/ml. What is the mass of 10 ml of aluminum? 4. Copper has a density of 8.9 g/ml. How much space does a 50 gram piece of copper take up? 5. Zinc has a density of 7.1g/mL. A piece of zinc has a volume of 60 ml. What is the mass of this piece of zinc? 6. A wood block has a length of 10cm, a width of 5 cm and a height of 2 cm. Its mass is 200 grams. What is its density?