PowerPoint 4.6 - FRQ Practice and Intermolecular Forces
advertisement

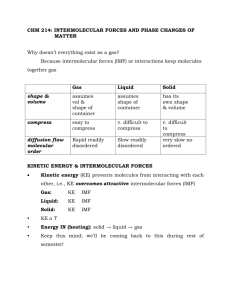
Catalyst End How To Approach FRQ 1. 2. 3. 4. Write down everything the problem gives you Identify what the question wants you to find Lay out a path to solve the problem Solve Free Response Practice Intermolecular Investigation • With your partners, complete the handout “Intermolecular Investigation” • Be sure to answer the questions when complete Lecture 4.5 – Solids, Liquids, and Intermolecular Forces Today’s Learning Targets • LT 4.8 – Using the kinetic molecular theory, I can explain the movement of atoms in liquids and solids. • LT 4.10 – I can compare and contrast the three types of intermolecular forces (Van der Waals, dispersion, and hydrogen bonding), identify the types in a given solution, and analyze chemical properties that result because of these forces. Kinetic Molecular Theory: Solids and Liquids • Recall, KMT states we can ignore the attractive forces between molecules in the gas phase. • We cannot ignore these forces in solids/liquids • As molecules cool, kinetic energy decreases and intermolecular forces increase Intermolecular Forces • Intermolecular Forces are the forces that exist between molecules • Much weaker than ionic, covalent, and metallic forces • Many properties result because of it Dispersion/London Forces • In an neutral atom, electrons are spread evenly throughout a molecule • When two molecules are brought close enough to one another, electrons rearrange to create a temporary dipole • The greater the polarizability of the molecule determines the strength of the dispersion force ▫ Increase as the molecular weight increases Dipole – Dipole Forces • When a molecule has a permanent dipole, it has attractive forces to other dipoles • Dipole – Dipole Forces are the attractive forces between molecules with partial positive and partial negative charges. • The larger the molecular weight, the larger the dipole – dipole force Hydrogen Bonding • When H bonds to N, O, or F, there is a huge difference in electronegativity • Hydrogen bonding is the intermolecular force between the hydrogen bound to N, O, or F and another N, O, or F atom. • Type of dipole – dipole interaction Ion – Dipole Forces • When we have a polar molecule, it can also be attracted to an ion. • This is an ion – dipole force Identifying Forces in Molecules • All molecules have London/dispersion forces ▫ Larger molecules have larger dispersion forces • The general strength of forces in molecules is: Dispersion < Dipole – Dipole < Hydrogen Bonding < Ion - Dipole Class Example • List the substance in order of increasing intermolecular forces: BaCl2, H2, CO, HF, and Ne. Table Talk • Order the molecules in order of increasing intermolecular forces CH3CH3, CH3OH, CH3CH2OH Properties From Intermolecular Forces: Boiling Point/Melting Point • The boiling point and melting point are dependent upon the amount of intermolecular forces • The more forces, the higher the boiling point and melting point because the molecules feel a greater attraction to one another. • This means more energy is necessary so that these forces can be broken. Properties From Intermolecular Forces: Viscosity • Viscosity is the resistance of a liquid to flow. • The more intermolecular forces, the higher the viscosity of the material Meet the Jesus Lizard Properties From Intermolecular Forces: Surface Tension • Surface Tension is the measure of the force it takes to break the surface of a given liquid • The more forces, the greater the surface tension due to the extensive intermolecular bonding Table Talk • Order the following molecules from lowest to highest boiling points: CH3CH2OH, CH3CH2CH2OH, CH3CH2CH2CH2OH, and HOCH2CH2OH Relay Races Relay Race Questions • What are the intermolecular forces present in SO2? • What are the intermolecular forces present in CH3COOH? • What are the intermolecular forces present in H2S? • Which substance has a larger dispersion force: H2S or H2O? • Which molecule has more forces: HF or C2H6? Closing Time • Read 11.1 – 11.3 and answer the essential questions • Do book problems: