22.3 – Writing formulas and Naming Compounds
advertisement

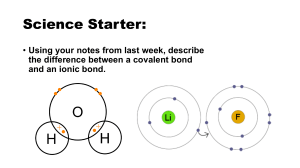
22.3 – Writing formulas and Naming Compounds Objectives Explain how to determine oxidation numbers Write formulas and names for ionic compounds Binary Ionic Compounds • A compound composed of 2 elements • To write a chemical formula for a binary compound, you need to know: • which elements are involved • how many electrons they lose or gain to become stable Oxidation Numbers Transition Elements • Because they can have multiple oxidation numbers, we have to express that number in the name with a Roman Numeral • For example, the oxidation number of iron in iron (III) oxide is 3+. Write Formulas for these Potassium ion Magnesium ion Copper (II) ion Chromium (VI) ion Barium ion Mercury (II) ion Ionic Compounds are NEUTRAL • Although the individual ions in a compound carry charges, the compound itself is neutral • A formula must have the right number of positive ions and the right number of negative ions so the charges balance Writing Formulas Write the formula for calcium chloride. Calcium is Ca+2 Chloride is Cl-1 Ca+2 Cl-1 would have a +1 charge. Need another Cl-1 Ca+2 Cl 1- Cl 1CaCl2 Step By Step – Writing Formulas 1. Write the symbol of the element (or polyatomic ion) with the positive oxidation number or charge 2. Write the symbol of the element (or polyatomic ion) with the negative oxidation number or charge 3. The charge (without the sign) of one ion becomes the subscript of the other ion. • Reduce the subscripts to the smallest whole numbers that retain the ratio of ions. Criss-Cross Method Polyatomic Ions • Groups of atoms that stay together and have a charge • A positively or negatively charged, covalently bonded group of atoms • Polyatomic ions as a whole contains two or more elements Polyatomic Formulas When writing out the formula for polyatomics, treat the polyatomic as a whole and place () around the symbol Then place any subscripts outside of the () because the subscript applies to the entire polyatomic YOU NEVER CHANGE THE FORMULA OF THE POLYATOMIC Name ammonium acetate chlorate chlorite cyanide hydroxide nitrate carbonate sulfate phosphate Symbol NH4+1 C2H3O2-1 ClO3-1 ClO2-1 CN-1 OH-1 NO3-1 CO3-2 SO4-2 PO4-3 Write the formulas for these Lithium sulfide Tin (II) oxide Tin (IV) oxide Magnesium fluoride Copper (II) sulfate Iron (III) phosphide Gallium nitrate Iron (III) sulfide Barium nitrate In-Class Assignment/Homework 22.3a Criss Cross WS 22.3a Criss-Cross practice