Alcohol Summary
advertisement

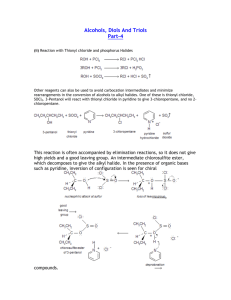
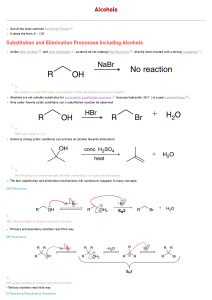
8 Important Concepts 1. Alcohols are alkanols (IUPAC) – Names derived from stem prefixed by alkyl and halo substituents Alcohols Have Polar and Short O-H Bond – 2. • • • • Hydroxy group is hydrophilic (hydrogen bonding) Unusually high boiling points Appreciable water solubility Alkyl part is hydrophobic 3. Alcohols Are Amphoteric – • • • • Deprotonation by bases whose conjugate acids are weaker than the alcohol Protonation yields alkyloxonium ions Acidity: primary > secondary > tertiary alcohol Electron-withdrawing substituents increase acidity 8 Important Concepts 4. Reverse Polarization – i.e., conversion of the alkyl group in a haloalkane, Cδ+-Xδ-, into its nucleophilic analog in an organometallic compound, Cδ--Mδ+. 5. Aldehyde and Ketone Carbonyl Carbons are Electrophilic – C=O carbon is subject to attack by hydride hydrogens or organometallic alkyl groups. Aqueous workup yields alcohols. 6. Oxidation of Alcohols – Yields aldehydes and 7. ketones (Chromium IV reagents). Retrosynthetic Analysis – Identifies efficient sequence of reactions by identifying strategic bonds.