Matter test Review 2010
advertisement

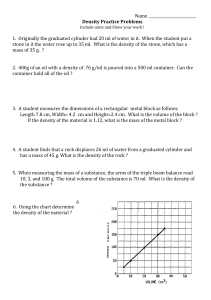
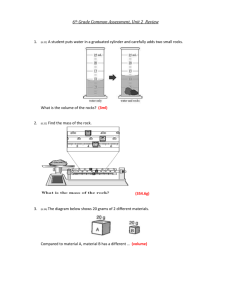
Matter Unit Review 1. Why is density important to scientists? 2. What are the four states of matter? 3. Where do you read the volume when reading a graduated cylinder? a. What volume is indicated in this picture? 4. If you let ice sit out on the counter, which state change results? 5. When you put water in a freezer, which state change occurs? 6. When you boil water, which state change occurs? 7. When dew forms on the grass after a cool night, what state change occurs? 8. Which units would be used to measure the mass of a person? 9. What has mass and takes up space? 10. When more than one type of atom bonds together, what is formed?. 11. A graduated cylinder had 21 mL of water in it. I put a rock in it and the volume went up to 37 mL. What is the volume of the rock? 12. What is the method, where you see how much the water rises in the graduated cylinder, of finding volume called? 13. What is the definition of density? 14. What is the definition of volume? 15. What is the definition of mass? 16. What is the temperature at which a substance starts to vaporize called? 17. What is the temperature at which a substance starts to change from a solid to a liquid called? 18. Which units would be best to describe the density of a solid bar of tungsten? 19. Which units would be best to describe the mass of a solid bar of titanium? 20. Which units would be best for describing the volume of water used in an experiment? 21. Which units would be best to describe the volume of a solid bar of nickel? 22. Which state of matter has molecules that are very far apart and move very fast? 23. Which state of matter has a defined volume, takes the shape of its container and has molecules or atoms that move freely? 24. Which state of matter has a defined shape and volume and has molecules or atoms that vibrate in place? Use the pictures below to answer questions 25-29. 25. Read the balance. What is the mass of this block? 26. What is the formula used to calculate the volume of a rectangular block? 27. The block in the picture above has a width of 3 cm, a length of 5 cm, and a height of 3 cm. What is the volume of this block? 28. What is the formula for calculating the density of an object? 29. Using your answers from questions number 25 and 27, calculate the density of the block.