Chemical Bonding I
advertisement

Chapter 9
Lewis Theory of Chemical Bonding
Lewis Bonding Theory
Emphasizes valence electrons to explain bonding
Lewis structures - Electron Dot Structures
Lewis structures allow us to predict many properties of
molecules - molecular stability, shape, size, polarity
Why Do Atoms Bond?
A chemical bond forms when the potential energy of
the bonded atoms is less than the potential energy of
the separate atoms.
To calculate this potential energy, you need to consider
the following interactions:
nucleus–to–nucleus repulsions
electron–to–electron repulsions
nucleus–to–electron attractions
Types of Bonds
Types of Atoms
Type of Bond
metals to
nonmetals
Ionic
nonmetals to
nonmetals
Covalent
metals to metals
Metallic
Bond
Characteristic
electrons
transferred
electrons
shared
electrons
pooled
Types of Bonding
Ionic Bonds
Metal atoms lose an electrons and become
cations.
Nonmetal atoms gain electrons and become
anions.
The oppositely charged ions are then
attracted to each other, resulting in an
ionic bond.
Covalent Bonds
Nonmetal atoms have relatively high
ionization energies, so it is difficult to
remove electrons from them.
When nonmetals bond together, it is
better in terms of potential energy for
the atoms to share valence
electrons.
The potential energy is lowest when the
electrons are between the
nuclei.
Atoms held together because shared
electrons are attracted to both
nuclei.
Metallic Bonds
The relatively low
ionization energy of metals
allows them to lose electrons
easily.
Metal atoms release their
valence electrons to be
shared as a pool by all the
atoms/ions in the metal.
An organization of metal
cation islands in a sea of
electrons.
Bonding results from
attraction of cation for
the delocalized electrons.
Valence Electrons & Bonding
Because valence electrons are held most loosely, and
Because chemical bonding involves the transfer or
sharing of electrons between two or more atoms,
Valence electrons are the most important in
bonding.
Determining the Number of
Valence Electrons in an Atom
The column number on the Periodic Table will tell you
how many valence electrons a main group atom has.
IA
IIA
IIIA
IVA
VA
VIA VIIA VIIIA
Li
Be
B
C
N
O
F
Ne
1e-
2e-
3e-
4e-
5e-
6e-
7e-
8e-
Lewis Structures of Atoms
We represent the valence electrons of main-group elements
as dots surrounding the symbol for the element.
IA
H
VIIIA
IIA
III
A
IVA
VA
VIA
VIIA
He
Li
Be
B
C
N
O
F
Ne
Na
Mg
Al
Si
P
S
Cl
Ar
Practice – Write the Lewis structure for
Arsenic
••
• As •
•
A lithium ion
A fluoride ion
Stable Electron Arrangements
and Ion Charge
Main-group ions and the noble gas configurati
Metals form cations by losing
enough electrons to get the same
electron configuration as the
previous noble gas.
Nonmetals form anions by
gaining enough electrons to get the
same electron configuration as the
next noble gas.
The noble gas electron
configuration must be very
stable.
Lewis Bonding Theory ⇒ Octet Rule
When atoms bond, they tend to gain, lose, or share
electrons to result in eight valence electrons
ns2np6 (noble gas configuration)
Exceptions
H, Li, Be, B attain an electron configuration like He
He = two valence electrons (a duet)
Li loses its one valence electron
H may share or gain one electron
It commonly loses its one electron to become H+
Be loses two electrons to become Be2+
It commonly shares its two electrons in covalent bonds, resulting
in four valence electrons
B loses three electrons to become B3+
It commonly shares its three electrons in covalent bonds,
resulting in six valence electrons
Expanded octets for elements in Period 3 or below
Lewis Theory and Ionic Bonding
Lewis symbols can be used to represent the
transfer of electrons from metal atom to
nonmetal atom, resulting in ions that are
attracted to each other and therefore
bond.
Sodium Chloride
Formation
Lewis Theory Predictions for
Ionic Bonding
Lewis theory predicts the number of
electrons a metal atom should lose or a
nonmetal atom should gain.
This allows us to predict the formulas of
ionic compounds that result.
It also allows us to predict the relative
strengths of the resulting ionic bonds
from Coulomb’s Law.
Predicting Ionic Formulas
Using Lewis Symbols
Electrons are transferred until the metal loses all its
valence electrons and the nonmetal has an octet.
Numbers of atoms are adjusted so the electron
transfer comes out even.
Li2O
Use Lewis theory to predict the chemical
formula of calcium chloride
· Cl ··
· Cl ··
·· ·· ·· ··
·
·
Ca
Ca2+
CaCl2
Use Lewis symbols to predict the formula of an ionic
compound made from reacting a metal, M, that has two valence
electrons with a nonmetal, X, that has five valence electrons
3M2+
M3X2
2X3Sr3N2
Energetics of Ionic Bond Formation
The ionization energy of the metal is endothermic:
Na(s) → Na+(g) + 1 e ─ "
ΔH° = +496 kJ/mol
The electron affinity of the nonmetal is exothermic:
½Cl2(g) + 1 e ─ → Cl─(g)" ΔH° = −244 kJ/mol
Therefore the formation of the ionic compound should be
endothermic.
Na(s) + ½Cl2(g) → NaCl(s)"
ΔH°f = + “something”
But the heat of formation of most ionic compounds is
exothermic and generally large.
Na(s) + ½Cl2(g) → NaCl(s)"
ΔH°f = −411 kJ/mol Why?
Ionic Bonding & the Crystal Lattice
The extra energy that is released comes
from the formation of a structure in which
every cation is surrounded by anions.
This structure is called a crystal lattice.
The crystal lattice is held together by
electrostatic attractions.
The crystal lattice maximizes these
attractions between cations and anions,
leading to the most stable arrangement.
Crystal Lattice
Electrostatic attraction is nondirectional!!
There is no direct anion–cation pair “bond”
Therefore, there is no ionic molecule.
The chemical formula for an ionic compound is
an empirical formula, simply giving the ratio of
ions based on charge balance.
Lattice Energy
The extra stability that accompanies the formation of
the crystal lattice is measured as the lattice
energy.
The lattice energy is the energy released when the
solid crystal forms from separate ions in the gas
state
1) Always exothermic
2) Can be calculated from knowledge of other processes
Lattice energy depends directly on size of charges
and inversely on distance between ions.
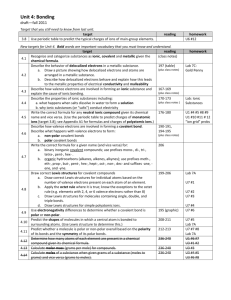
Practice – Given the information below,
determine the lattice energy of NaCl
Na(s) → Na(g) +108 kJ
½ Cl2(g) → Cl(g) +½(244 kJ)
Na(g) → Na+(g) +496 kJ
Cl (g) → Cl−(g) −349 kJ
Na(s) + ½ Cl2(g) → NaCl(s) −411 kJ
?
Na+ (g) + Cl−(g) → NaCl(s) ΔH (NaCl lattice)
Determining Lattice Energy
The Born–Haber Cycle
The Born–Haber Cycle is a hypothetical series
of reactions that represents the formation of an
ionic compound from its constituent elements.
The reactions are chosen so that the change in
enthalpy of each reaction is known except for
the last one, which is the lattice energy.
Naº (s) + ½ Cl2 (g)
NaCl (s)
Born–Haber Cycle
for NaCl
ΔH°f (metal atoms, g)
separating atoms
ΔH°f (nonmetal atoms, g)
ΔH°f (salt)
ΔH°f (cations, g)
forming ions
ΔH°f (anions, g)
ΔH°(crystal lattice)
forming lattice
Born–Haber Cycle
Use Hess’s Law to add up enthalpy changes of other
reactions to determine the lattice energy.
ΔH°f(salt) = ΔH°f(metal atoms, g) + ΔH°f(nonmetal atoms, g)
+ ΔH°f(cations, g) + ΔH°f(anions, g)
+ ΔH°(crystal lattice)
ΔH°f(NaCl, s) =
ΔH°f(Na atoms,g)
ΔH°f(NaCl, s) =
ΔH°f [Na(s)--->Na(g)] (Heat of vaporization)
+ ΔH°f(Cl atoms,g)
+ ΔH°f (Cl–Cl bond energy)
+ ΔH°f(Na+,g)
+ Na 1st Ionization Energy
+ ΔH°f(Cl−,g)
+ Cl Electron Affinity
+ ΔH°(NaCl lattice)
+ NaCl Lattice Energy
Na(s) → Na(g) +108 kJ
½ Cl2(g) → Cl(g) +½(244 kJ)
Na(g) → Na+(g) +496 kJ
Cl (g) → Cl−(g) −349 kJ
Na+ (g) + Cl−(g) → NaCl(s) ΔH (NaCl lattice)
Na(s) + ½ Cl2(g) → NaCl(s) −411 kJ (measured in an experiment)
NaCl Lattice Energy =
ΔH°f(NaCl, s)
− [ΔH°f(Na atoms,g) + ΔH°f(Cl–Cl bond energy)
+ Na 1st Ionization Energy + Cl Electron Affinity]
NaCl Lattice Energy = (−411 kJ)
− [(+108 kJ) + (+122 kJ)
+ (+496 kJ) + (−349 kJ) ]
= −788 kJ
Practice – Given the information below,
determine the lattice energy of MgCl2
Mg(s) ➔ Mg(g)
ΔH1°f = +147.1 kJ/mol
½ Cl2(g) ➔ Cl(g)
ΔH2°f = +122 kJ/mol
Mg(g) ➔ Mg+(g)
ΔH3°f = +738 kJ/mol
Mg+(g) ➔ Mg2+(g) ΔH4°f = +1450 kJ/mol
Cl(g) ➔ Cl−(g)
ΔH5°f = −349 kJ/mol
Mg(s) + Cl2(g) ➔ MgCl2(s) ΔH6°f = −641 kJ/mol
Practice – Given the information below,
determine the lattice energy of MgCl2
Mg(s) ➔ Mg(g)
2{½ Cl2(g) ➔ Cl(g)}
Mg(g) ➔ Mg+(g)
Mg+(g) ➔ Mg2+(g)
2{Cl(g) ➔ Cl−(g)}
ΔH1°f = +147.1 kJ/mol
2ΔH2°f = 2(+122 kJ/mol)
ΔH3°f = +738 kJ/mol
ΔH4°f = +1450 kJ/mol
2ΔH5°f = 2(−349 kJ/mol)
−
2+
Mg (g) + 2 Cl (g) ➔ MgCl2(s)
ΔH° lattice energy = ? kJ/mol
Mg(s) + Cl2(g) ➔ MgCl2(s)
ΔH6°f = −641 kJ/mol
Trends in Lattice Energy
Ion Size
The force of attraction between charged
particles is inversely proportional to the
distance between them.
Larger ions mean the center of positive charge
(nucleus of the cation) is farther away from
the negative charge (electrons of the anion).
larger ion = weaker attraction
weaker attraction = smaller lattice energy
Lattice Energy vs.
Ion Size
Trends in Lattice Energy
Ion Charge
The force of attraction between oppositely charged particles is
directly proportional to the product of the charges.
Larger charge means the ions are more strongly attracted.
larger charge = stronger attraction
stronger attraction = larger lattice energy
Of the two factors, ion charge is generally more important
Lattice Energy =
−910 kJ/mol
Lattice Energy =
−3414 kJ/mol
Lattice Energies of Some Ionic Solids (kJ/mole)
-- - →
++ +
23+
2+
2+
2(M
+
X
(2M
X
M2X)
(M
+
X
MX3)
(M
(M + 2X
→
MX2)
MX)
3X →
→ MX)
M2X3)
Anions
Cations
F-
Cl-
Br-
I-
O2-
Li+
1036
853
807
757
2,925
Na+
923
787
747
704
2,695
K+
821
715
682
649
2,360
Be2+
3,505
3,020
2,914
2,800
4,443
Mg2+
2,957
2,524
2,440
2,327
3,791
Ca2+
2,630
2,258
2,176
2,074
3,401
Al3+
5,215
5,492
5,361
5,218
15,916
Order the following ionic compounds in order
of increasing magnitude of lattice energy:
CaO, KBr, KCl, SrO
First examine the ion charges and order by sum of the charges
(KBr, KCl) < (CaO, SrO)
Then examine the ion sizes of each group and order by radius
larger < smaller
KBr < KCl < SrO < CaO
Order the following ionic compounds in order of
increasing magnitude of lattice energy:
MgS, NaBr, LiBr, SrS
First examine the ion charges and order by sum of the charges
(NaBr, LiBr) < (MgS, SrS)
Then examine the ion sizes of each group and order by radius
larger < smaller
NaBr < LiBr < SrS < MgS
Ionic Bonding-Model vs. Observations
Lewis theory implies strong attractions between ions.
Lewis theory predicts high melting points and boiling
points for ionic compounds.
The stronger the attraction (larger the lattice energy),
the higher the melting point.
Ionic compounds have high melting points and
boiling points (MP generally > 300 °C).
All ionic compounds are solids at room
temperature.
Ionic Compounds Melt
Melting an ionic solid
Ionic Bonding-Model vs. Observations
Lewis theory implies that the positions of the ions
in the crystal lattice are critical to the stability of
the structure
Lewis theory predicts that moving ions out of
position should therefore be difficult, and ionic
solids should be hard
Ionic solids are relatively hard
(compared to most molecular solids)
Ionic Bonding-Model vs. Observations
Lewis theory implies that if the ions are displaced from
their position in the crystal lattice, that repulsive forces
should occur
This predicts the crystal will become unstable and break
apart. Lewis theory predicts ionic solids will be brittle.
Ionic solids are brittle. When struck they shatter.
Ionic Bonding-Model vs. Observations
To conduct electricity, a material must have charged
particles that are able to flow through the material
Lewis theory implies that, in the ionic solid, the ions are
locked in position and cannot move around
Lewis theory predicts that ionic solids should not
conduct electricity
Ionic solids do not conduct electricity
Conductivity of NaCl
in NaCl(s), the
ions are stuck in
position and not
allowed to move
to the charged
rods
Ionic Bonding-Model vs. Observations
Lewis theory implies that, in the liquid state or when
dissolved in water, the ions will have the ability to
move around
Lewis theory predicts that both a liquid ionic compound
and an ionic compound dissolved in water should
conduct electricity
Ionic compounds conduct electricity in the
liquid state or when dissolved in water
Conductivity of NaCl
in NaCl(aq),
the ions are
separated and
allowed to
move to the
charged rods