Activity Series of Elements Reference Sheet
advertisement

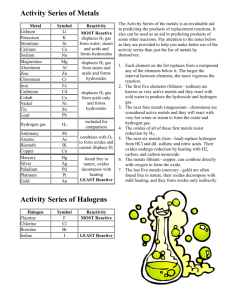
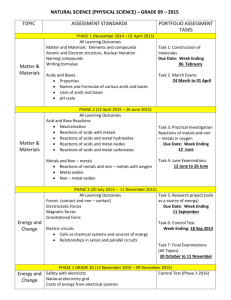
Activity Series of the Elements The Activity Series of the metals is an invaluable aid to predicting the products of replacement reactions. It also can be used as an aid in predicting products of some other reactions. Pay attention to the notes below as they are provided to help you make better use of the activity series than just the list of metals by themselves. 1. Each element on the list replaces from a compound in aqueous solution any of the elements below it. The larger the interval between elements, the more vigorous the reaction. 2. The first seven elements (lithium - sodium) are known as very active metals and they react with cold water to produce the hydroxide and hydrogen gas. 3. The next five metals (magnesium - chromium) are considered active metals and they will react with very hot water or steam to form the oxide and hydrogen gas. 4. The oxides of all of these first metals resist reduction by H2. 5. The next six metals (iron - lead) replace hydrogen from HCl and dil. sulfuric and nitric acids. Their oxides undergo decomposition by heating with H2, carbon, and carbon monoxide. 6. The metals lithium - copper, can combine directly with oxygen to form the oxide. 7. The last five metals (mercury - gold) are often found free in nature, their oxides decompose with mild heating, and they form oxides only indirectly Metal Reactivity Lithium Rubdium Potassium Barium Strontium Calcium Sodium Magnesium Aluminum Manganese Zinc Chromium Iron Cadmium React with cold Water and Acids, replacing hydrogen. React readily with Oxygen forming oxides. React with Steam(Not Cold H2O) and Acids, replacing hydrogen. React readily with oxygen forming Oxides The metal oxides below decompose in presence of heat & hydrogen Cobalt Nickel Tin Lead React with Acids but not water to replace hydrogen React readily with oxygen forming Oxides Hydrogen Antimony Arsenic Bismuth Copper React readily with oxygen. forming Oxides Mercury Silver Palladium Platinum Gold Mostly non-reactive Decompose upon heating alone NonMetal Halogen Reactivity F2 Cl2 Br2 I2 Anion above replaces Anion below on list.