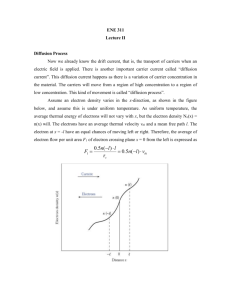
Eq. 1
advertisement

Lecture 3 MSE 515 Solutions of Schrodinger equation for special cases Last time: −ħ2 𝜕Ψ 2 ∇ 𝜓 + 𝑉𝜓 = 𝐸𝜓 = 𝑖ℏ 𝜕𝑡 2𝑚 Ψ 𝑖𝑠 𝑐𝑎𝑙𝑙𝑒𝑑 𝑎 𝑤𝑎𝑣𝑒𝑓𝑢𝑛𝑐𝑡𝑖𝑜𝑛 +∞ ∫ |Ψ(𝑥)|2 𝑑𝑥 = 1 −∞ The probability of finding it must be unity. This is a critical point and will come up over and over. 1. Free electrons: (1-D) (Simple but very important one) - We consider electrons that propagate freely in potential free space in the positive x-direction 2 - ∇ 𝜓 → 𝑑2 𝜓 𝑑𝑥 2 - −ħ2 𝑑2 𝜓 2𝑚 𝑑𝑥 2 = 𝐸𝜓 _____ Eq. 1 - 𝜓(𝑥 ) = 𝑒 𝑖𝛼𝑥 1 - 𝑑𝜓 𝑑𝑥 = 𝑖𝛼𝑒 𝑖𝛼𝑥 - 𝑑2 𝜓 𝑑𝑥 2 = 𝑖 2 𝛼 2 𝑒 𝑖𝛼𝑥 - In Eq. 1 ħ2 2 𝑖𝛼𝑥 𝛼 𝑒 = 𝐸𝜓 2𝑚 - ħ2 𝛼 2 𝑖𝛼𝑥 𝑒 = 𝐸𝜓 𝑟𝑒𝑚𝑒𝑚𝑏𝑒𝑟 𝜓 = 𝑒 𝑖𝛼𝑥 2𝑚 ħ2 𝛼 2 ∴𝐸= 2𝑚 -Since no boundary conditions, all values of energy are allowed. α =√ 2𝑚𝐸 ħ2 ______Eq. 2 𝑝2 = 𝐸(𝐾. 𝐸. ) 𝑝 (𝑚𝑜𝑚𝑒𝑛𝑡𝑢𝑚) 2𝑚 𝑝2 = 2𝑚𝐸 𝑖𝑛 𝐸𝑞. 1. α=√ 𝑝2 ħ2 2 𝑝 =ħ = α= 2𝜋 𝜆 𝑝 ∙ 2𝜋 ℎ ′ → 𝜆 𝑑𝑒 𝐵𝑟𝑜𝑔𝑙𝑖𝑒 𝑠 ℎ𝑦𝑝𝑜𝑡ℎ𝑒𝑠𝑖𝑠 𝑝 ℎ =𝑘 = k (wave number) K is a vector → 𝑘𝑥 𝑘𝑦 𝑘𝑧 λ→ wave vector Note: k – vector describes the wave properties of an electron. Quantum Mechanics (k) Classical Mechanism (p) 𝑝 And remember k = ħ Back to Energy E, we can find ħ2 E = 2𝑚 𝑘 2 ψ(x) = 𝑒 𝑖𝑘𝑥 Separation of variables ψ(x,t) = 𝑒 𝑖𝑘𝑥 ∙ 𝑒 𝑖𝜔𝑡 This is an equation of traveling wave. It represents a free particle. 2. Bound electrons [ potential wall] 3 - We consider that the electron can move freely between infinitely high potential barriers Electrons cannot escape because of the potential wall Note: The potential inside the wall is zero. The difference between this case and last case is the boundary conditions. −ħ2 𝑑 2 𝜓 = 𝐸𝜓 2𝑚 𝑑𝑥 2 Assume the solution is ψ(x) = AeiαL + Be-iαL 4 𝛼= √ 2𝑚𝐸 ħ2 Applying Boundary Conditions at x = zero ψ = 0 at x = L ψ = 0 since ψ = 0 , x ≤ 0 , x ≥ L 0 = A𝑒 𝑧𝑒𝑟𝑜 + 𝐵𝑒 𝑧𝑒𝑟𝑜 A = -B or B = -A A𝑒 𝑖𝛼𝑎 − 𝐴𝑒 −𝑖𝛼𝑎 = 0 A (𝑒 𝑖𝛼𝑎 − 𝑒 −𝑖𝛼𝑎 ) = 0 A EULER'S FORMULA IS THE KEY TO UNLOCKING THE SECRETS OF QUANTUM PHYSICS From Euler’s Equation 1 𝑖𝛿 sin 𝛿 = (𝑒 − 𝑒 −𝑖𝛿 ) 2𝑖 2𝑖𝐴 𝑖𝛼𝐿 (𝑒 − 𝑒 −𝑖𝛼𝐿 ) = 0 2𝑖 2A* 𝑖 sin (α L) = 0 5 ∴ sin α L = 0 → αL = n𝜋 α= 𝑛𝜋 n= 0, 1, 2, 3... (integer) 𝐿 from α = √ 2𝑚𝐸 ħ2 ħ2 E = 2𝑚 𝛼 2 ħ2 𝑛 2 𝜋 2 = 2𝑚 𝑎𝐿2 ħ2 𝜋 2 E = 2𝑚𝑎𝐿2 𝑛2 n = 1, 2, 3… 6 - The second case is similar to an electron bound to its atomic nucleus - Only certain energy levels are allowed for the electron - This is “Energy quantization” - Probability of finding the electron at inside the well: 𝜓𝑛 = A (𝑒 𝑖𝛼𝑥 − 𝑒 −𝑖𝛼𝑥 ) na = 2𝐴𝑖 ∙ sin 𝛼𝑥 𝜓𝜓 ∗ = 4𝐴2 𝑠𝑖𝑛2 𝛼𝑥 7 3. Finite potential barrier: (Tunnel effect) Assume a free electron propagating in the positive xdirection meets a potential barrier V0 (higher than the total energy of electron). 8 Write sch. Eq. For each region: Region I. V=0 𝑑2 𝜓 𝑑𝑥 2 + 2𝑚 ħ2 𝐸𝜓 Region II. 𝑑2 𝜓 𝑑𝑥 2 + 2𝑚 ħ2 (𝐸 − 𝑉𝑜 )𝜓 = 0 Region I solutions: 𝜓𝐼 = 𝐴𝑒 𝑖𝑎𝑥 + 𝐵𝑒 −𝑖𝑎𝑥 9 𝑎= √ 2𝑚𝐸 ħ2 Region II solution: 𝜓𝐼𝐼 = 𝐶𝑒 𝑖𝛽𝑥 + 𝐷𝑒 −𝑖𝛽𝑥 Only certain solutions exist (for which n is integer) Let’s plot the energy solutions for the two cases: 10 The second case is similar to an electron bond to a nucleus. 11 First three levels of the stationary state and probability of those states. Region I solutions: 𝜓𝐼 = 𝐴𝑒 𝑖𝑎𝑥 + 𝐵𝑒 −𝑖𝑎𝑥 𝑎= √ 2𝑚𝐸 ħ2 Region II solution: 𝜓𝐼𝐼 = 𝐶𝑒 𝑖𝛽𝑥 + 𝐷𝑒 −𝑖𝛽𝑥 2𝑚 β = √ ħ2 (𝐸 − 𝑉0 ) (E – V0) is less than zero 12 β → imaginary = if 2𝑚 ∴ 𝛾 = √ ℎ2 (𝑉0 − 𝐸 ) 𝜓𝐼𝐼 = 𝐶𝑒 𝛾𝑥 + 𝐷𝑒 −𝛾𝑥 Using boundary conditions X→ ∞ 𝜓𝐼𝐼 = 𝐶 ∙ ∞ + 𝐷 ∙ 0 C must be zero ∴ 𝜓𝐼𝐼 = 𝐷𝑒 −𝛾𝑥 Ψ decreases in region II exponentially The decrease is higher for larger , for larger potential barrier 13 14 The electron wave propagates in the finite potential barrier. Tunneling effect: penetration of a potential barrier - This is only quantum mechanical effect. - In classical mechanics: If the electron kinetic energy is smaller than V, the electron will be entirely reflected and “cannot overcome the barrier” Examples of tunneling : - Tunneling of electrons from one metal to another through an oxide film. 15 - Emission of alpha particles from nuclei by tunneling through the binding potential barrier. 4. Another Case - We can find that electron can penetrate region II and propagates in region III. 16 −ħ2 2 𝜕Ψ ∇ 𝜓 + 𝑉𝜓 = 𝐸𝜓 = 𝑖ℏ 2𝑚 𝜕𝑡 Partial differ eq. in 2 - ∇ 𝜓 → - = h / 2p Y 𝑑2 𝜓 𝑑𝑥 2 𝑑2 𝜓 + 𝑑𝑦 2 + 𝑑2 𝜓 𝑑𝑧 2 mass = m V = potential |𝜓(𝑥, 𝑦, 𝑧, 𝑡)|2 dx dy dz gives the probably of finding the electron in volume dx dy dz −ħ2 ∇2 𝜓 2𝑚 𝜓 1 𝛿𝜔 + 𝑉 = 𝑖ħ 𝜔 function r 𝛿𝑡 Eq. 1 function time 𝜓(𝑟, 𝑡) = 𝜓(𝑟)𝜔(𝑡) Eq 2 Separation of variables substitute Eq 2 in Eq. 1 17 For each Eq. to be correct it must be equal to a constant - For each equation to be right, it must equal to constant −ħ2 ∇2 𝜓 2𝑚 𝜓 𝜕𝜔 +𝑉 =𝐸 iħ 𝜕𝑡 = 𝐸𝜔 E is a constant → Eq. 3 E is the same const. solution: 𝜔 = exp[ −𝑖𝐸𝑡 ħ ] from eq. 3 −ħ2 ( 2𝑚 ∇2 + 𝑉) 𝜓 = 𝐸𝜓 time independent Schrödinger Equation. It will be applied to be calculations of stationery conditions. 18