Metal Reactivity with Acids: Lesson Plan
advertisement

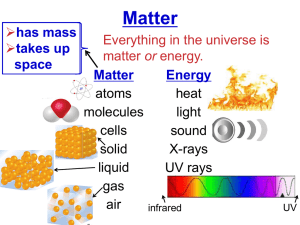
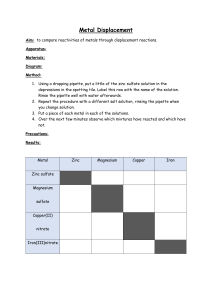
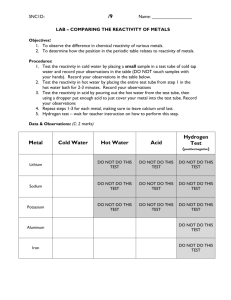
Lesson 7 Reactions of metals with acids. ILO's to be able to recall that hydrogen and a salt are produced when a metal reacts with an acid to be able to describe observations and draw conclusions about relative reactivity from them to decide which observations and measurements are appropriate to deduce relative reactivity Activities 1) Pupils carry out test tube reactions with 1M hydrochloric acid. Test tubes should be half-filled with the acid and the metals added in small quantities. Magnesium should be used one strip at a time. They will need to be patient with Al as it takes some time for the oxide layer to disappear, if it does. Pupils may test for the gas hydrogen using the pop test if you choose, depending upon the ability of the group. Metals used should be the same as for the last lesson with the exception of Ca! Again pupils should draw up their own list of relative reactivity, using the results of the water and acid experiments, including all the metals from potassium to copper. 2) Bring the class together and ask for their version of the reactivity series and then compare it with the real thing. K Na Ca Mg Al Zn Fe Sn Pb Cu You may wish to show silver and gold as being less reactive than copper at this point. Resources Activity 1 Resources: Lesson 7 Class set Al, Cu, Pb pieces Fe filings, Zn granules Mg in 2cm strips 1M Hydrochloric acid (6 bottles) Sieves Matches Homework Write a mnemonic for the reactivity series (Like 'Richard Of York Gave Battle In Vain' sort of thing)