Metals Reactivity Lab
advertisement

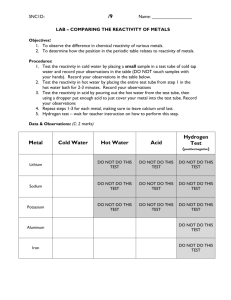
Name(s):________________________________ Period: ________ Metals Reactivity Lab Purpose: To investigate the properties of the metals and their reactivity. Procedure: 1. Take a small scoop of Mg and add it to a beaker with 20 ml of water. After one minute test the resulting solution with red litmus. Record your observations. 2. Repeat step # 1 with Ca. 3. Repeat step #1 with Al. 4. Take a small scoop of Mg in a beaker to 5 ml of 2M HCl. Record your observations. 5. Repeat step #4 with Ca. 6. Repeat step #4 with Al. Observations: Include observations for all 6 reactions, including the results of the litmus tests Li Write the reactions in the boxes corresponding to their relative position in the periodic table. Na Mg K Ca Al Discussion: 1. Which group is the most reactive? Least reactive? 2. Why was this group the most reactive? 3. Does reactivity increase or decrease down a group? Why? 4. Does reactivity increase or decrease across a period? Why? 5. How many valence electrons do the alkali metals have? Alkaline Earth? 6. Compare the reactivity of the six elements to water and create a theory about the trend in the periodic table for metals. 7. Why does your trend increase/decrease down a group or across a period? Be specific. 8. Does ionization energy effect reactivity? How would the 2 concepts overlap?