b-Dikarbonilvegyületek reakciói Qu1: How many a
advertisement

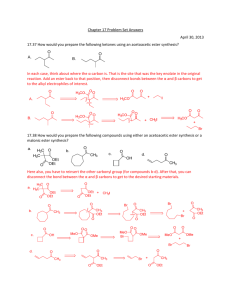
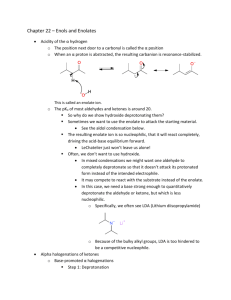
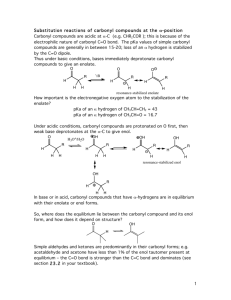
1 -Dikarbonilvegyületek reakciói Qu1: How many -hydrogens are there in each of the following ester systems ? (a) ethyl ethanoate (d) ethyl benzoate (b) methyl ethanoate (e) diethyl propanedioate (c) methyl methanoate Qu 2: What are the major products of the reactions when each of the following esters are treated with sodium ethoxide: (a) Ethyl ethanoate (b) Methyl benzoate (c) Ethyl propanoate (d) Diethyl hexanedioate (e) Diethyl heptanedioate (f) Ethyl 5-oxohexanoate Qu 3: What are the major products of the following reaction sequences ? (a) (b) (c) Qu 4: How would you prepare diethyl hexanedioate from cyclohexene ? Qu 5: How would you prepare ethyl acetoacetate (ie. ethyl 3-oxobutanoate) ? Qu 6: How would you prepare diethyl malonate from malonic acid ? 2 Qu. 7 Qu. 8 Qu. 9 3 Qu. 10 4 Qu. 11 C7H5NO4 MW = 167, 3-nitrobenzoesav 5 C8H6O2 MW = 134 tereftálaldehid 6 C10H12O MW = 148: butirofenon 7 8 C8H6O3 MW = 150, 5-hidroxi-3H-benzofurán-2-on 9 10 Megoldások Qu1: First step is can you draw the structures The -hydrogens are those on the carbon adjacent to the carbonyl group, in the first four examples shown below this is to the left, and in the fifth example between the two carbonyl groups. (a) (b) (c) 3 hydrogens as there is a methyl group attached to the carbonyl group. 3 hydrogens as there is a methyl group attached to the carbonyl group. No hydrogens as there is no carbon atom attached to the carbonyl group. No hydrogens as there carbon atom attached to the carbonyl group does not have a hydrogen attached to it. (d) (e) 2 hydrogens as there is a methylene group (-CH2-) between the 2 carbonyl groups. Qu.2 (a) This is the Claisen condensation reaction. An ester enolate nucleophile attacks a second molecule of the ester, displacing an ethoxide, that will be protonated giving the alcohol. (b) Transesterification converts the methyl ester to the ethyl ester. There is no condensation reaction since there are no -hydrogens. 11 (c) This is a Claisen condensation reaction. An ester enolate nucleophile attacks a second molecule of the ester, displacing the alcohol portion as a leaving group which will be protonated giving the alcohol. (d) This is an intramolecular Claisen known as a Dieckmann condensation. Numbering the chain helps avoid making the wrong size ring in the product (and keep track of any substituents if there are any). Form the enolate adjacent to one of the esters and attack the other. Here, the enolate at C5 is shown attacking the carbonyl at C1 giving the five membered ring. (e) This is an another Dieckmann condensation. Here, the enolate at C6 will attack the carbonyl at C1 giving the cyclohexane derivative. (f) This is a condensation of a ketone enolate with an ester. We should form the enolate adjacent to the ketone, either at C4 or C6. Look for the most stable product. Here, the enolate at C6 will attack the carbonyl at C1 giving the cyclohexane derivative in preference to a more strained cyclobutane derivative. Qu3: 12 (a) An example of an acetoacetic ester synthesis. Alkylation of an active methylene enolate followed by decarboxylation to give the ketone. (b) An example of an acetoacetic ester synthesis applied to a cyclic system. Alkylation of an active methylene enolate followed by decarboxylation to give the cyclic ketone. (c) First part of a malonic ester synthesis, alkylation of the active methylene enolate giving the alkylated diester. 13 Qu4: This question reviews some material from previous chapters..... Cyclohexene can be cleaved by either ozonolysis (or by oxidative cleavage of an diol). Oxidation of the dialdehyde gives the dicarboxylic acid which can be converted into the diester by Fischer esterification. Qu.5 The most likely method is via a Claisen condensation reaction of ethyl ethanoate. An ester enolate nucleophile attacks a second molecule of the ester, displacing an ethoxide, that will be protonated giving the alcohol. Qu. 6: Esters are typically made from the parent carboxylic acid by the Fischer esterification. Qu.7 14 Qu. 8