Assignment 3
advertisement

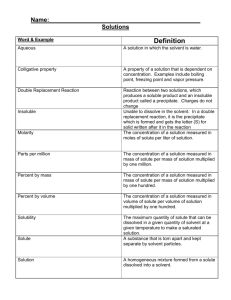
Name: ______________________ Date: ______________________ Science 14 Assignment #3: Mixtures and Their Uses 1. Which of the following is a mechanical mixture? A. shampoo B. chocolate chip cookie dough C. tap water D. air 2. Which of the following is a solution? A. concrete B. sand and water C. orange juice D. apple juice 3. Sugar and water are mixed to form a solution. The solute is A. sugar B. water C. both sugar and water D. none of the above 4. The solvent of a solution is defined as A. the non-dissolved particles B. the substance that dissolves C. the substance into which something is dissolved D. the substance that remains in a filter after the solution is filtered 5. You can recognize a solution because A. it is always colourless B. you can see the particles in it C. you can see only one type of substance D. it is always a solid 6. In a solution of 5 mL of baking soda and 250 mL of water, the solvent is ____________. 7. A substance that will dissolve in a solvent is said to be A. concentrated B. insoluble C. filtered D. soluble 8. A substance that is soluble in water is A. gasoline B. salt C. oil D. petroleum jelly 9. A substance that is insoluble in water is A. corn oil B. sugar C. baking soda D. salt 10. Which statement is true about solubility and temperature? A. The solubility of a solute decreases as temperature increases. B. The solubility of a solute increases as temperature increases. C. The solubility of a solute increases as temperature decreases. D. The solubility of a solute stays the same as temperature increases. Use the following graph to answer questions 11 to 13. 11. Which solute has the lowest solubility at 60°C? A. sodium chloride B. potassium chlorate C. ammonium chloride D. both A and B 12. Ammonium chloride has a solubility of about 400 g/L at A. 27°C B. 37°C C. 77°C D. 95°C 13. Which substance's solubility does not seem affected much by temperature change? A. ammonium chloride B. potassium chlorate C. sodium chloride D. both A and C 14. Soap helps remove grease from clothes because A. grease is insoluble in soap B. grease is soluble in water C. soap is not soluble in water D. grease is soluble in a soap and water solution 15. The concentration 12 g/L means there are 12 g of solute present in __________ of solution. 16. Concentration describes the amount of ____________________ present in a solution. 17. The process used to collect salt from a salt-water solution is called A. filtration B. distillation C. sedimentation D. crystallization 18. A process by which water can be cleaned by boiling and then collecting the steam is called A. filtration B. distillation C. sedimentation D. crystallization 19. A window screen can be fitted to the bottom of a box frame and used to separate which of the following? A. a solution B. a mechanical mixture C. a solute from a solvent D. bacteria from water 20. Which substance that sticks to bacteria, viruses, and tiny dirt particles, causing them to settle to the bottom, is used in water purification? A. chlorine B. sand C. alum D. fluorine 21. The air you breathe is a solution of about 78% nitrogen and 21% oxygen and 1% other gases. Which substance is the solvent? Explain how you know. ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ 22. The following steps are used to separate a mixture of sugar, sand, and nails. List the letter of steps in the order they should be completed. A. Use a filter to separate the sand from the solution. B. Separate the nails from the mixture using a magnet or sieve. C. Heat the solution to evaporate the water and crystallize the sugar. D. Mix water with the sand and sugar.