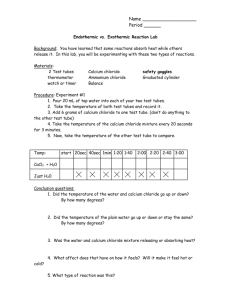
Physical Separation
Activity
1. Look up the following physical properties of ammonium chloride (NH4Cl), silicon dioxide
(SiO2, sand, quartz), and sodium chloride (NaCl, table salt).
Substance
Formula
sodium
chloride
NaCl
ammonium
chloride
NH4Cl
Melting Point (°C)*
Solubility†
Appearance
silicon
SiO2
dioxide
(Sand)
*Do not use the triple point (tp) for ammonium chloride; use the sublimation point (sp).
†
Solubility: soluble (s) or insoluble (i) in water
2. (4) A student is given a 3.589 g mixture of iron filings, calcium chloride and sand. She
separates the mixture and recovers 0.897 g of iron, 0.923 g of sand and 1.686 g of calcium
chloride. Calculate the percentage of each of the three components she recovered from the
original mixture and the percent of material she lost during the separation process.
Physical Separation
Activity
3. Using the above physical properties, complete the following flow chart by giving the
reagents and/or conditions necessary (in the ovals) to affect each indicated separation step and
how the components (in the boxes) will be separated.
Mixture of NaCl, SiO2, NH4Cl
 0
0