Electrons in Atoms Notes
advertisement

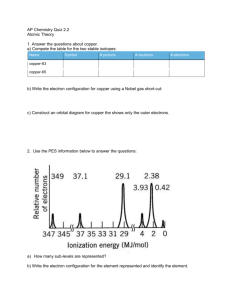
Name __________________________________ Guided Notes: Electrons in Atoms Draw an atom of lithium. How did scientists figure out the electron energy levels? Can they see them? _________ Question for the day: How did scientists figure out that electrons are in certain energy levels? Scientists got clues from ____________________. Atoms absorb and emit it in distinct ways. What does a prism do? A prism breaks light into its ________________________________. Sunlight has a continuous spectrum. Sketch a continuous spectrum here: A _____________ is another example of the continuous spectrum of sunlight. All the colors blend into each other. Every possible energy level of light is present. When scientists look at the light coming off a pure element, they don’t see a continuous spectrum, they see a ____________ spectrum. Sketch the line spectrum of hydrogen. But leave some space to add more to it later on. Every element has a unique line spectrum. Name an important application of this discovery. _______________________________________________________________________________________ As you know, the color of a light corresponds to its ______________________________. Atoms only emit light of certain ___________________________. But why don’t they emit light of all the energies? Neils Bohr came up with a radical answer. Electrons can orbit the nucleus only in certain allowed energy levels. Think of it like _________________ on a ladder. You can be on one rung, or another rung, but not really between rungs. Electrons can be in one ___________________________ or jump to another _______________________, but they’re not allowed to be in between the allowed energy levels. Electrons can’t be just anywhere. Name __________________________________ It takes energy to move an electron away from the nucleus. Why? _______________________________ Conversely, if an electron moves back closer to the nucleus, energy is ____________________________. Remember, energy is required to move an electron to a _________________ energy level, and energy is released when an electron moves to a ____________________ energy level. The fact that atoms give off characteristic colors of light (________________________) provides clues about how electrons are arranged in atoms. The line spectra gave rise to two important ideas: 1. Electrons exist only in certain _______________________________ around the nucleus. 2. Energy is ____________________________ when an electron moves to a higher level; energy is ________________________ when an electron moves to a lower level. Go back to the hydrogen line spectrum you drew earlier, and label each line with the energy change for the electron that it corresponds to. Copy the bohr model of the hydrogen atom below, showing the changes in energy that show up as colored lines in the line spectrum: