Chiral Drug Design
advertisement
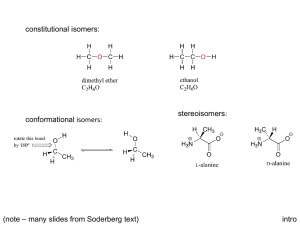
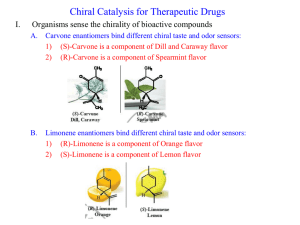
Chiral Drug Design History & Importance of Chiral Drug Design We’ve been talking about Stereoisomers…. • Isomers – Constitutional Isomers – Stereoisomers • • • • Cis vs trans (EZ) Enantiomers Diastereomers Meso Compounds (later next week) Molecules with Asymmetric Centers… An sp3 atom, usually carbon, with 4 UNIQUE groups attached! Asymmetric center = chiral center CH3 Cl H OH If a molecule has at least one chiral center… It can be an optically active molecule. -Enantiomers -Diastereomers *Meso compounds are optically INACTIVE. Optically Active… Rotates the plane of polarized light clockwise (+) or counterclockwise (-) (+/- have no bearing on the type of asymmetric center; we will explain types of chiral centers in a minute!) Chiral centers have… “handedness” Enantiomers are non-superimposable mirror images! Enantiomers - Chiral Centers are Complete Opposites (R/S, S/R, etc) - R/S determination after slide show! - Identical chemical and physical properties - Except rotation of plane-polarized light (S)-Alanine (left) and (R)-alanine (right) How are Diastereomers Different? - Have at least 2 chiral centers – partially opposite chirality. - Chiral centers are PARTIAL opposites - R,S : R, R - R, S : S, S - Have DIFFERENT chemical & physical properties. (-)Threose (-) Erythrose Importance For Drug Design • The body is chiral • Enantiomers of compounds can react differently in the body, with greatly helpful or harmful outcomes “Nature has a way of knowing how to make things work. Reactions often run in a catalytic mode, and material use, energy, and waste are minimized. Many molecules are chiral, and their unique handedness has both intricate and dramatic influences on how they interact with biological systems.” 1957: Thalidomide (Racemic Mixture) The two enantiomers of thalidomide: Left: (S)-thalidomide Right: (R)-thalidomide Thalidomide • German drugmaker Chemie Grünenthal introduced thalidomide, under the name Contergan, to the German market on Oct. 1, 1957 • It was a sedative to treat insomnia as well as to reduce nausea associated with pregnancy. • By 1960, the drug was in more than 20 countries in Europe and Africa. • On Nov. 18, 1961, the German paper Welt am Sonntag reported on a study finding that pregnant women who had been taking thalidomide were giving birth to babies with gross deformities. • "By November 27, Grünenthal had pulled the drug off the market, blaming the sensationalism of the press" Frances Oldham Kelsey • Joined the FDA in 1960 • Richardson-Merrell Inc. submitted a New Drug Application to market thalidomide in the U.S. under the brand name Kevadon. • At the time, the prevailing law was the 1938 Federal Food, Drug & Cosmetic Act • On March 8, 1962, Richardson-Merrell withdrew its drug application, after the effects were widely publicized. Frances helped uncover… The S-enantiomer of thalidomide, caused the teratogenic effects (body mutations etc). The once believed "safe" R-isomer can be converted to the teratogenic isomer once metabolized human body. 1938 Federal Food, Drug & Cosmetic Act • required proof of safety to be submitted to FDA before a drug could be approved for marketing • did not require demonstration of efficacy • allowed "experimental" use of drugs while approval was being sought, which meant that a drug could be distributed widely before it was approved Thalidomide sparked major changes for the FDA “the drug approval process was under considerable criticism, particularly the quality of the scientific data submitted in New Drug Applications and the lack of an efficacy requirement,’ according to Kelsey, writing in the 1996 annual report of FDA's Office of Compliance. ‘The nature and magnitude of the thalidomide disaster,’ she wrote, spurred the swift passage of legislation addressing the shortcomings of the 1938 law that had been sitting in Congress before the disaster. Known as Kefauver-Harris Drug Amendments, they were signed into law by President John F. Kennedy in October 1962.” Impact on Today • Old racemic mixture drugs are often reassessed as single enantiomers • Synthesis, separation and analysis of chiral compounds has advanced greatly, and is highly sought (chromatography) Some Statistics… • 2006, 80% of small-molecule drugs approved by the FDA were chiral • 75% were single enantiomers • About 200 chiral compounds should enter the market each year • The field of asymmetric synthesis is expanding rapidly, 50% of chiral drugs use chiral technologies • Due to FDA policy as of 1994, decision to create racemic mixture must be justified in quality, safety, and efficacy, may be case-by-case Sepracor • • • • Located in Marlborough, MA Founded in 1984, first major drug in 2000 Lunesta, Xopenex, Xopenex HFA, Brovana Schering-Plough for CLARINEX; Sanofi-Aventis for ALLEGRA; and UCB Pharma for XYZAL /XUSAL™. • Focus on treatment of respiratory and central nervous system disorders • Insomnia, asthma, chronic obstructive pulmonary disease, depression Levalbuterol • XOPENEX is (R)-enantiomer of albuterol • short-acting bronchodilator for reversible obstructive airway disease, such as asthma. Levalbuterol caused fewer reported side effects than albuterol. Effects: Levalbuterol vs albuterol Tremor, 22% vs 78%. Nervousness, 0% vs 56%. Palpitations, 0% vs 56%. Tachycardia, 0% vs 44%. Conclusion Chiral drugs are very important, in terms of efficacy and applications in the human body, but they are not a universal solution, and there are still many risks in their development. “Nature has a way of knowing how to make things work. Reactions often run in a catalytic mode, and material use, energy, and waste are minimized. Many molecules are chiral, and their unique handedness has both intricate and dramatic influences on how they interact with biological systems.” • So let’s continue practicing!!!! – Drawing our Perspective & Fischer (bow-tie on a skelton) Projections – Identifying type of Chirality