Optical Isomers Terms and Concepts
advertisement

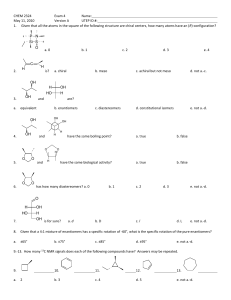
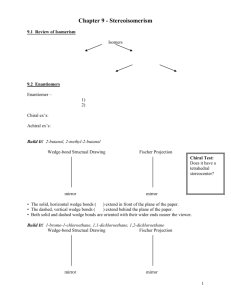
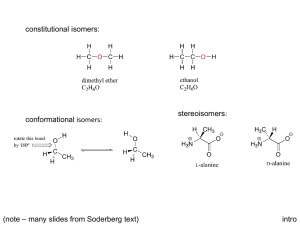
Optical Isomers Terms and Concepts 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Chiral – asymmetrical Achiral – symmetrical Chiral carbon – asymmetrical carbon by being bonded to 4 different groups Optically active – ability to rotate plane polarized light – all chiral molecules are optically active however you need a polarimeter in order to determine the direction and angle the isomer rotates light a. dextrorotary – rotates plane polarized light clockwise [ά] = (+) b. levarotary – rotates plane polarized light counterclockwise [ά] = (-) Enantiomers – non super imposable mirror images – only chiral molecules can have enantiomers a. opposite configuration (R vs. S) at all chiral centers b. identical physical properties (ie. Boiling point, density etc) – difficult to separate – the separation process is called resolution c. rotate light in equal but opposite directions Racemic mixture – an optically inactive solution that has 1:1 ratio of enantiomers Enantiomeric excess – an optically active solution that contains an uneven ratio of enantiomers Diastereomers – stereoisomers that are not enantiomers – not limited to chiral molecules like enantiomers a. cis/trans or E/Z b. partially opposite configuration – minimum of one chiral center for each isomer must have identical configuration (if applicable) c. different physical properties – can be separated physically for example via distillation d. there is no relationship as to how they rotate light with respect to each other Meso molecule – achiral molecules that contain chiral centers a. optically inactive b. has diastereomers but don’t have an enantiomer How to assign R vs. S configuration a. Prioritize the 4 groups bonded to the chiral center b. If group #4 is not on a wedge or dash rotate and redraw the molecule until group #4 is on a wedge or dash c. Draw an arrow from #1 to #2 to #3 – arrow may pass through #4 d. Assign configuration based on direction of arrow and orientation of #4 #4 on a dash #4 on a wedge Clockwise Counterclockwise R S S R