Covalent Bonds - Research at OSU Chemistry
advertisement

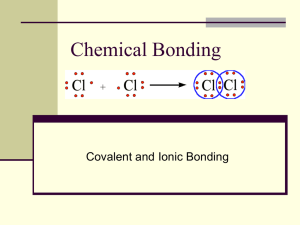
Chapter 6: Inorganic and Organic Compounds 6.5 Chem 1110 Figures: Basic Chemistry 3rd Ed., Timberlake and Timberlake Ionic vs. Covalent Bonding Ionic Bonding Covalent Bonding • Metal - nonmetal (NaCl) • Electron transfer and electrostatic interactions • Formula Unit (from larger arrays) • Solids • Two nonmetals (O2, H2O) • Electron sharing in bonds • Dissolve in water and conduct electricity • Discrete molecules • All physical states (solid, liquid and gas) • If they dissolve in water, they do not conduct electricity The Covalent Bond Covalent Bonding: a chemical bond that results from two nuclei attracting the same shared electron pair Forming a H2 Molecule • A covalent bond forms as two H atoms move close together to share electrons Diatomic Elements Several elements share electrons to form diatomic, covalent molecules: Octet Rule Covalent compounds also obey the Octet Rule: • Each atom in the bond is seeking a noble gas configuration • Electrons are shared to achieve the octet • For Example: Br → needs 1 electron to achieve an octet Lewis Dot Structures • We can use Lewis Dot structures to help us form Covalent Bonds: H with Cl H with O P with Br Electron-Dot Formulas Electron-dot formulas: • the order of bonded atoms in a covalent compound • the bonding pairs of electrons between atoms • the unshared (lone) valence electrons • a central atom with an octet Covalent Compounds Bonding and Lone Pair Electrons • Bonding pair of electrons are shared to create the covalent bond • Non-bonding pair or lone pair make up the octet Bonding and Lone Pair Electrons • Bonding pair of electrons are shared to create the covalent bond • Non-bonding pair or lone pair make up the octet Cl2 HBr H2S Naming Covalent Compounds 1) Most metallic atom comes first then the more negative + “-ide” 2) Name includes the total number of each atom: For Example: NO NO2 N2O Table 6.10 (p. 178) Naming Covalent Compounds For Example: SiBr4 P4O10 Naming Covalent Compounds • Many covalent compounds have common names that you are most likely familiar with: NH3 H2O H2S CH4 Formulas and Names of Some Covalent Compounds Learning Check Select the correct name for each compound: A. SiCl4 1) silicon chloride 2) tetrasilicon chloride 3) silicon tetrachloride B. P2O5 1) phosphorus oxide 2) phosphorus pentoxide 3) diphosphorus pentoxide C. Cl2O7 1) dichlorine heptoxide 2) dichlorine oxide 3) chlorine heptoxide Learning Check Write the name of each covalent compound: CO _____________________ CO2 _____________________ PCl3 _____________________ CCl4 _____________________ N2O _____________________