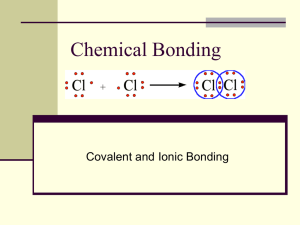
Covalent Bonds
advertisement

Covalent Bonds By Wendy Bradley Acacia Dunphy Andrew Hudson Kaitlyn Tanner Properties of covalent compounds A single covalent bond is formed when a pair of electrons is shared between two atoms Usually have lower melting points and boiling points They are more flammable than ionic compounds They don’t conduct electricity Usually aren’t soluble in water. There are two kinds of covalent bonds; polar and non-polar (pure) covalent bonds. Electronegativity The tendency of an atom to draw or attract the electrons in a bond toward itself. To form a covalent bond, two or more atoms with similar electronegativities will share electrons Electronegativity is like a game of tug-of-war with a puppy. An atom's ability to pull determines what kind of bond it forms. Electronegativity Trends Lewis Structure Also known as electron-dot diagram, which is a simple diagram showing the valence electrons as dots. Some diagrams show the bonding between different atoms Gilbert N. Lewis came up with the idea of the octet rule, which is that atoms react by changing the number of their electrons, so as to acquire the stable electron structure of a noble gas. Resonances Occurs when two or more equally valid electron dot diagram can be written for a molecule Earlier chemists thought that the electron pairs were rapidly flipping back and forth or resonating between the various electron dot diagrams But more recently we have found that electron pairs do not resonate. The actual resonance bonding is considered to be hybrid or a mixture of the extremes represented by the resonance forms. Used to represent certain types of molecular structures A very important part in the valence bond theory Polar covalent compounds When two different atoms are joined by a covalent bond and the bonding electrons are shared unequally. Also called ‘Polar Bond’ for short Page 336 and 324 in the white/green book Interactive Bonding Lab Each of the following activities is done in groups of 2-3 people. Purpose: to be able to visualize bonding, angles, multiple bonds, as well as the position of lone pairs in molecular formulas. Materials: Large Marshmallows, small colored marshmallows, red-hot candies, gumdrops, and toothpicks Compounds involved: H2O-Water CH4--Methane C2H4-Ethane N2-Diatomic Nitrogen CH2O-Formaldehyde NH3-Ammonia Procedure: Make Water Molecule O-large marshmallow H-small marshmallow Lone pair-red-hot candies Make Diatomic Nitrogen Molecule N-Large Marshmallows Lone Pairs-Red-hot candies Make Formaldehyde Molecule C-Large Marshmallow H-Small Marshmallows O-Large Gumdrop Red-hot candies if needed ***Remember to use more toothpicks if multiple bonds occur. Across 3. These special electrons are found in the outer most shell of an atom 5. _____ bonds are bonds formed when two different atoms are joined by covalent bond and the bonding electrons are shared equally 6. All atoms want to be happy or ______ 8. Gilbert N. Lewis came up with the _______ rule 9. A substance that can be separated into simpler substances (elements or other compounds) only by chemical reactions 10. The force between electrons and protons 11. When two or more electron-dot diagrams can be written for a molecule Down 1. The tendency of an atom to draw or attract the electrons in a bond toward itself 2. ____________ bonds 4. Also known as the electron-dot diagram 5. A non-polar covalent compound 7. _________ you will find in orbitals