lec15_print
advertisement

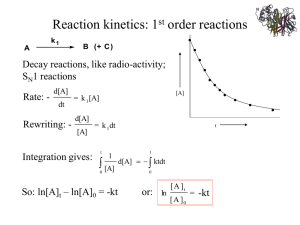
Lecture 15 Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors in which they take place. Today’s lecture Enzymes Michealis-Menten Kinetics Lineweaver-Burk Plot Enzyme Inhibition Competitive Uncompetitive 2 Last lecture 3 Last lecture 4 Enzymes Michaelis-Menten Kinetics. Enzymes are protein like substances with catalytic properties. Enzyme unease. [From Biochemistry, 3/E by Stryer, copywrited 1988 by Lubert Stryer. Used with permission of W.H. Freeman and Company.] 5 Enzymes It provides a pathway for the substrate to proceed at a faster rate.The substrate, S, reacts to form a product P. S Slow P ES Fast A given enzyme can only catalyze only one reaction. Example, Urea is decomposed by the enzyme urease. 6 7 8 A given enzyme can only catalyze only one reaction. Urea is decomposed by the enzyme urease, as shown below. 2O NH2CONH2 UREASE H 2NH3 CO2 UREASE 2O S E H PE 9 The corresponding mechanism is: E S E S k1 E S E S k2 E S W P E k3 10 Michaelis-Menten Kinetics rP k 3 E SW rES 0 k1 ES k 2 E S k 3WE S k1 E S E S k 2 k 3W E t E E S 11 Et E k1S 1 k 2 k 3W Michaelis-Menten Kinetics Vmax k 3 W E tS k catE t S rP k 3 E SW k 2 k 3W K S m S k1 k cat Km VmaxS rP k 3 E SW Km S 12 Vmax=kcat* Et Turnover Number: kcat Number of substrate molecules (moles) converted to product in a given time (s) on a single enzyme molecule (molecules/molecule/time) For the reaction kcat H2O2 + E →H2O + O + E 40,000,000 molecules of H2O2 converted to product per second on a single enzyme molecule. 13 Summary 14 Michaelis-Menten Equation VmaxS rP rS KM S (Michaelis-Menten plot) Vmax Vmax VmaxS1/ 2 2 K M S1/ 2 Solving: -rs KM=S1/2 S1/2 15 CS therefore KM is the concentration at which the rate is half the maximum rate Inverting yields 1 1 KM 1 rS Vmax Vmax S Lineweaver-Burk Plot 1/-rS slope = KM/Vmax 1/Vmax 16 1/S Types of Enzyme Inhibition Competitive E I I E (inactive) Uncompetitive E S I I E S (inactive) Non-competitive E S I I E S (inactive) 17 I E S I E S (inactive) Competitive Inhibition 18 Competitive Inhibition k3 k1 E S E S EP k2 k4 E I E I ( inactive ) k5 1) Mechanisms: E S E S E S P E EI E I rP k 3C ES 19 E S E S E I EI Competitive Inhibition 2) Rates: rES 0 k1CSCE k 2CES k 3CES k1CSC E CSC E C ES k 2 k3 Km k 3CSCE rP Km rIE 0 k 4CICE k 5CIE 20 C I E CICE KI k5 KI k4 Competitive Inhibition C Etot C E C ES C IE k 3C EtotCS rP CI K m K m CS KI VmaxCS rS CI CS K m 1 KI 21 1 1 k m CI 1 1 rS Vmax Vmax K I CS C Etot CE CS C I 1 Km KI Competitive Inhibition From before (no competition): 1 1 Km 1 rS Vmax Vmax CS Increasing CI Competitive No Inhibition Competitive 1 rS slope Intercept 22 1 Vmax Km Vmax 1 1 K m CI 1 1 rS Vmax Vmax K I CS 1 CS Intercept does not change, slope increases as inhibitor concentration increases 23 Uncompetitive Inhibition Inhibition only has affinity for enzyme-substrate complex E S k1 k2 k3 E S P k4 I E S I E S (inactive) k5 Developing the rate law rP rS k cat E S rES 0 k1 ES k 2 E S k cat E S k 4 IE S k 5 I E S (1) rIES 0 k 4 IE S k 5 I E S 24 (2) Adding (1) and (2) k1 E S k 2 E S k cat E S 0 k1 E S E S E S k 2 k cat KM From (2) k4 I E S I E S I E S IE S k5 KI K IK M k5 KI k4 25 k cat E S rp k cat E S KM Total enzyme E t E E S I E S S IS E 1 K M K I K M rp 26 k cat E t S S IS K M 1 K K K M I M Vmax S rS rP I K M S1 KI I 1 1 K M S1 rS Vmax S K I 1 K M 1 1 I 1 rS Vmax S Vmax K I Slope remains the same but intercept changes as inhibitor concentration is increased 27 Lineweaver-Burk Plot for uncompetitive inhibition 28 Noncompetitive Inhibition (Mixed) E+S +I E·S -I -I (inactive)I.E + S P+E +I I.E.S (inactive) Increasing I 1 rS No Inhibition Both slope and intercept changes 1 CS 29 rS VmaxCS CI k m CS 1 kI 1 1 C I k m 1 C I 1 1 rS Vmax k I Vmax CS k I 30 End of Lecture 15 31