E.Q. Why is it important to know the different

E.Q.
Why is it important to know the different parts of an atom?
All notes must be in the student’s own handwriting.
Chunk the information on the right to form questions in this column.
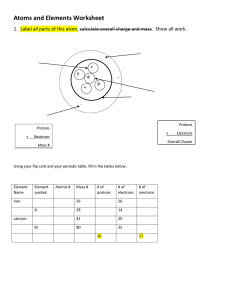
Atoms
Matter is anything that takes up space & has mass.
Matter can be solid, liquid, gas, or plasma.
Mass is the amount of matter an object is made of.
Atoms are the smallest particle an element can be divided into and still remain the same substance.
Atoms are made of 3 types of subatomic particles but consist mostly of empty space.
Nucleus
the center of the atom
largest part and therefore has most of the mass
consists of protons and neutrons
Protons
in the nucleus
positively charged
determines the name of the element (same as atomic number)
Neutrons
in the nucleus
neutral charge (no charge) electron cloud is the region surrounding the nucleus
Electrons
in the cloud
negatively charged
so small they virtually have no mass
Shells are the rings we draw around the nucleus.
The first shell can hold up to 2 electrons.
The second shell can hold up to 8 electrons.
The last shell is the valence shell.
Summary
Write a summary about the parts of atoms here. Make sure to use complete sentences and write a full paragraph, not just a few sentences.