8-32 (i.e., reversible-adiabatic).
advertisement

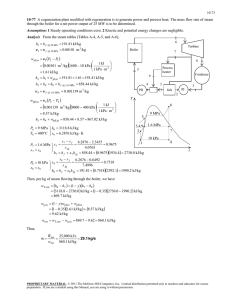
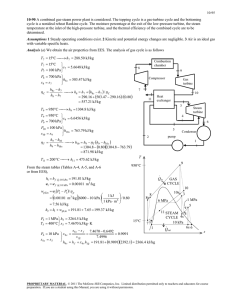
8-32 8-60 Steam is expanded in an isentropic turbine. The work produced is to be determined. Assumptions 1 This is a steady-flow process since there is no change with time. 2 The process is isentropic (i.e., reversible-adiabatic). Analysis There is one inlet and two exits. We take the turbine as the system, which is a control volume since mass crosses the boundary. The energy balance for this steady-flow system can be expressed in the rate form as E E in out Rate of net energy transfer by heat, work, and mass E in m 1 h1 W out 'E system Ê0 (steady) 0 4 MPa 5 kg/s Rate of change in internal, kinetic, potential, etc. energies Steam turbine E out m 2 h2 m 3 h3 W out 700 kPa m 1 h1 m 2 h2 m 3 h3 50 kPa 100°C From a mass balance, m 2 0.05m 1 (0.05)(5 kg/s) 0.25 kg/s m 3 0.95m 1 (0.95)(5 kg/s) 4.75 kg/s T Noting that the expansion process is isentropic, the enthalpies at three states are determined as follows: P3 T3 50 kPa ½ h3 ¾ 100qC ¿ s 3 P1 4 MPa s1 s3 P2 700 kPa s2 s3 2682.4 kJ/kg 7.6953 kJ/kg K 1 4 MPa 0.7 MPa 50 kPa (Table A - 6) ½ ¾ h1 7.6953 kJ/kg K ¿ 3979.3 kJ/kg (Table A - 6) ½ ¾ h2 7.6953 kJ/kg K ¿ 3309.1 kJ/kg 2 3 s (Table A - 6) Substituting, W out m 1 h1 m 2 h2 m 3 h3 (5 kg/s)(3979.3 kJ/kg) (0.25 kg/s)(3309.1 kJ/kg) (4.75 kg/s)(2682.4 kJ/kg) 6328 kW PROPRIETARY MATERIAL. © 2008 The McGraw-Hill Companies, Inc. Limited distribution permitted only to teachers and educators for course preparation. If you are a student using this Manual, you are using it without permission.