6.1 Types of Energy
advertisement

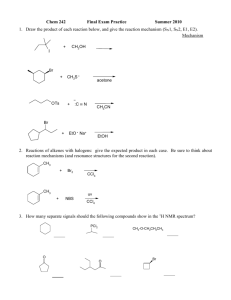
Announcements & Agenda (04/04/07) Will start with quiz today Review session today @ 3pm (same place) Today Amides (13.5) Sugars! Section 14.1-14.3 Carbohydrates Monosaccharides Cyclic Monosaccharides (?) 1 Last Time: Hydrolysis of Esters • an ester reacts with water to produce a carboxylic acid and an alcohol. • an acid catalyst is required. O H+ H—C—O—CH2—CH3 + H2O O H—C—OH + H—O—CH2—CH3 Base Hydroylsis forms an alcohol plus the salt of a carboxylic acid! 2 What acid and alcohol make the following ester? O H 3C 0% 1. 2. 3. 4. 0% 0% 0% 1 C 2 3 4 O C H2 C H2 CH3 Ethanol (2C) and acetic acid (2C) Propanol (3C) & acetic acid (2C) Ethanol (2C) & propionic acid (3C) Methanol (1C) & propionic acid (3C) 5 3 Last Time: Base Hydrolysis of Triglycerides Make Soaps 4 Last Time: Amines (Derivatives of NH3) Typically 3 kinds: • primary (1°) amines: 1 carbon group is bonded to N • secondary (2°) amines: 2 carbon groups bonded to N • tertiary (3°) amines: 3 carbon groups bonded to N. Can H-bond like alcohols, although N is not as polar • decent BPs & solubility in water Act as weak bases: CH3—NH2 + H2O CH3—NH3+ + OH– Useful properties of amine salts: • solids at RT, very soluble in body fluids (ideal for drug molecules, and salts don’t stink 5 Last Time: Alkaloids • physiologically active nitrogen-containing compounds. • often addictive. N CH 3 nicotine 6 Morphine and Codeine • alkaloids. • obtained from the oriental poppy plant. • used as painkillers. • modified to make heroin. 7 Chocolate Chocolate is found in cocoa beans O H CH3 N N O N N CH3 8 Last Time: Amides (13.5) made from… Amine + Carboxylic Acid Amides are produced • by reacting a carboxylic acid with ammonia or an amine (1° or 2°). • using heat. O Heat CH3—C—OH + NH3 O CH3—C—NH2 + H2O O O Heat CH3—C—OH + CH3—NH2 CH3—C—NH—CH3 + H2O 9 Some Amides in Health and Medicine 10 Physical Properties of Amides SIMILAR TO AMINES… • primary (−NH2) or secondary (−NH−) amides form hydrogen bonds. • primary have higher mp’s than secondary. • tertiary (no H on N) do not form hydrogen bonds & have lower melting points. • all form hydrogen bonds with water. • with 1-5 carbon atoms are soluble in water. 11 Hydrolysis of Amides IMPORTANT TO KNOW Amides undergo • Acid hydrolysis to produce a carboxylic acid and an ammonium salt. • Base hydrolysis to produce the salt of a carboxylic acid and an amine or ammonia. • REACTIONS LOOK SIMILAR TO THOSE OF ESTERS!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!! 12 Hydrolysis Reactions acid hydrolysis O || CH3—C—NH2 O || CH3—C—OH + NH4+Cl– HCl + H2O NaOH O || CH3—C—O– Na+ + NH3 base hydrolysis 13 Ch 14 Carbohydrates • a major source of energy from our diet • composed of C, H, & O • also known as saccharides, which means “sugars.” 14 Practice Problems (Ch 14) 14.05, 14.07, 14.09, 14.13, 14.15, 14.19, 14.21, 14.23, 14.25, 14.27, 14.29, 14.31, 14.33, 14.35, 14.37, 14.39, 14.41, 14.47, 14.49, 14.51, 14.53, 14.57, 14.59 15 Carbohydrates • produced by photosynthesis in plants • Example: glucose synthesized in plants from CO2, H2O, and energy from the sun. • are oxidized in living cells to produce CO2, H2O, and energy. 16 Types of Carbohydrates • Monosaccharides: simplest carbohydrates. • Disaccharides: consist of two monosaccharides. • Polysaccharides: contain many monosaccharides. 17 Monosaccharides • typically 3-6 carbon atoms • have a C=O group (aldehyde or ketone) • • aldoses ketoses • several hydroxyl groups 18 Aldoses • monosaccharides with an aldehyde group… • …and many hydroxyl groups. triose (3C atoms) tetrose (4C atoms) pentose (5C atoms) hexose (6C atoms) O ║ C─H aldose │ H─ C─OH │ H─ C─OH │ CH2OH Erythose 19 Ketoses CH2OH • monosaccharides with a │ ketone group… C=O ketose • …and many hydroxyl │ groups. H─ C─OH │ H─ C─OH │ H─C─OH │ Fructose, a ketohexose CH2OH 20 Fischer Projections • also used to represent carbohydrates. • places the most oxidized group at the top. • shows chiral carbons as the intersection of vertical and horizontal lines. 21 D & L Notations: Know This Now… In a Fischer projection, the −OH group on the • chiral carbon farthest from the carbonyl group determines an L or D isomer. • left = L for the L-form. • right = D for the D-form. (MOST COMMON IN NATURE!) 22 Learning Check Identify each as the D or L isomer. A. B. C. CH2OH O C H HO H HO H H HO H HO CH2OH L __-Ribose O O C H HO H OH H OH H H OH CH2OH L Threose __- CH2OH D Fructose __23 D-Glucose • found in fruits, corn syrup, and honey • an aldohexose with the formula C6H12O6 • known as blood sugar in the body • the monosaccharide in polymers of starch, cellulose, and glycogen 24 Blood Glucose Level In the body, • normal blood: [glucose] = 70-90 mg/dL. • a glucose tolerance test measures blood glucose for several hours after ingesting glucose. 25 D-Fructose • is a ketohexose C6H12O6 • is the sweetest carbohydrate • found in fruit juices and honey • converts to glucose in the body CH2OH C O HO C H H C OH H C OH CH2OH D-Fructose 26 D-Galactose • is an aldohexose C6H12O6. • not found free in nature. • is obtained from lactose, a disaccharide. • has a similar structure to glucose except for the –OH on C4 Which C is C4??? O C H H C OH HO C H HO C H H C OH CH2OH D-Galactose 27 Cyclic Structures (14.3) • prevalent form of monosaccharides with 5 or 6 carbon atoms O O • form when the hydroxyl group on C-5 reacts with the aldehyde group or ketone group • Orgo-Chem reaction not previously mentioned: “C=O” + ROH → hemiacetal 28 Drawing Cyclic Structures: Practice! Example: Glucose STEP 1 Number the carbon chain and turn clockwise to form a linear open chain. H O C 1 H 2 C OH HO 3 C H H 4 C OH H 5C OH H H OH H O HOCH2 C C C C C 6 5 4 3 2 1 H OH OH H OH 6 CH OH 2 29 Cyclic Structure for Glucose STEP 2 Fold into a hexagon. • Bond the C5 –O– to C1. • Place the C6 group above the ring. • Write the –OH groups on C2 & C4 below the ring (These are the C atoms that pointed down from Step 1). • Write the –OH group on C3 above the ring. • Write a new –OH on C1. CH2OH 6 5 O 4 OH 1 OH 3 2 OH OH 30 Cyclic Structure for Glucose STEP 3 Write the new –OH on C1 • down for the form. • up for the form. CH2OH O OH OH O OH OH -D-Glucose CH2OH OH OH OH OH -D-Glucose 31 Summary of the Formation of Cyclic Glucose 32 Mutarotation • cyclic structures open and close. • -D-glucose converts to β-D-glucose & vice versa. • at any time, only a small amount of open chain forms. CH2OH CH2OH O OH H O OH OH OH OH -D-glucose (36%) CH2OH O O C OH H OH D-glucose (open) (trace) OH OH OH OH β-D-glucose (64%) 33 Cyclic Structure of Fructose • is a ketohexose. • reacts the -OH on C-5 with the C=O on C-2 CH2OH C O CH2OH HO C H CH2OH OH H C OH H C OH CH2OH O O OH OH OH CH2OH OH OH α-D-fructose -D-fructose CH2OH D-fructose 34