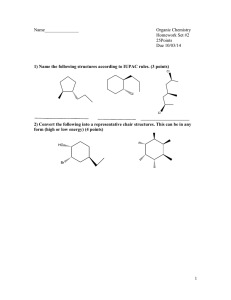
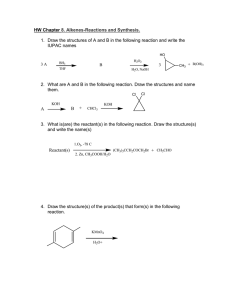
1.c 2.b 3.c 4.a 5.butane, 2 + 13 yield 8 + 10 CO is poisonous to
advertisement

1.c 2.b 3.c 4.a 5.butane, 2 + 13 yield 8 + 10 CO is poisonous to humans; hydrocarbons hurt to Ozone add Br2 to water or burn; A would not change color or burn light, B color change burns darker CH3CHCHCH3 + HBr yields CH3CHBrCH2CH3; addition 6. change/ replacement or atom/group by speices with a non-bonding/ lone pair of electron deficient of a molecule CH3-CH2-CH2-CH2Br/ CH2-CH-CH2Br CH3 CH3-CH2-CHBr-CH3 CH3 CH3 – C – Br CH3 H R – C – Cl H Cl2 yield 2Cl. Initiation Cl. + C6H5CH3 yields C6H5CH2 + HCl Propagation C6H5CH2Cl termination Or just write curly arrow for direction for all electrons To break the Cl-Cl bond 8. Same empirical formula, differ by CH2 ethanoic acid is higher; larger mass, more intermolecular forces, larger surface area any OH located on a different carbon