biological_molecules
advertisement

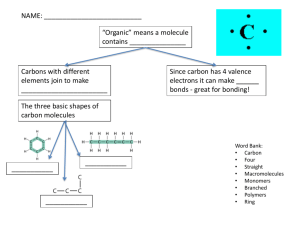
Food, glorious food All about biological molecules Four basic biological molecules Carbohydrates Proteins Lipids Nucleic Acids (Which we will look at later) Monomers and Polymers “Mono” means “one.” “Poly” means “many.” Large biological molecules are made up of smaller ones. Carbohydrates Sugars: the basic building blocks of complex carbohydrates. Polysaccharides: long chains of sugars. Some include: starch cellulose (“fiber”) glycogen Sugars Sugars are the simplest carbohydrates. Sugars can be single-ring (monosaccharides) or double-ring (disaccharides). Glucose, a monosaccharide Notice the ringshaped structure. Glucose is an energy source for all eukaryotic organisms. Your brain uses 30% of all glucose you consume. Maltose, a disaccharide Notice the two-ring structure. Anything look familiar? Maltose is made up of two glucose molecules. Forming Maltose Notice that we’re about to lose an H from one glucose and an OH from the other. Both glucose molecules will link to one oxygen atom. Because water (HOH) is lost, this is called dehydration synthesis. Sucrose, a disaccharide Note the components: glucose and fructose. Starch Starch is a polysaccharide A starch molecule is a long chain of hundreds of glucose molecules. Starch Here is a model of starch (also called amylose). Notice that dehydration synthesis happens here, also. Cellulose Here is a model of cellulose. What do you see here that was different in starch? The difference is small, but crucial. Starch and Cellulose See it now? That’s all it takes to make starch digestible and cellulose (“fiber”) indigestible to humans. Lipids Lipids are fats, waxes, and oils. Lipids are made up of fatty acid chains and glycerine molecules. Lipid molecule structure The head of a lipid molecule may be polar, attracting water. Fatty acids, being nonpolar, repel water. Saturated vs. Unsaturated A side-by-side comparison of a saturated (hydrogenated) and a monounsaturated fatty acid. Saturated vs. Unsaturated Saturated fatty acids pack together neatly, making fats that are solid at room temperature. Saturated vs. Unsaturated The bent chains of unsaturated fats do not fit together neatly. Unsaturated fats are liquid at room temperature. Thinking question Shortening and margarine labels often brag that their products are made with healthy, unsaturated vegetable oils. So why are they solid at room temperature? Hint: Check the ingredients. What does “hydrogenated” mean? Proteins Egg whites are made up of a protein called albumin. Like all proteins, albumin is composed of thousands of amino acids. Protein Proteins are polymers. Proteins are long chains of amino acids. Amino Acids An amino acid has three critical parts: amine group carboxyl (acid) group R group (side chain) Proteins As you can see, it takes many, many amino acids to make up a protein such as albumin. Proteins Amino acids link together with peptide bonds. As you can see, peptide bonding is a type of dehydration synthesis. An amino acid chain folds to form a protein. Two types of proteins: Structural proteins are used to build fibers and tissues. Enzymatic proteins take part in chemical processes. Recap Starches and other polysaccharides are long chains of sugars. Proteins are made up of amino acids. Lipids (fats, waxes, oils) are made up of fatty acids and glycerol molecules.