Document
advertisement

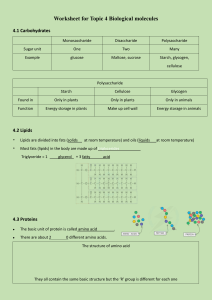
Chemical composition of cells Lab 3 Introduction Inorganic molecules nonliving things Organic molecules living organisms Salt molecules: NaCl (Sodium chloride Na+ Cl-) Sugar Organic compounds in living thing • • • • Proteins Carbohydrates Lipids Nucleic acids Concepts • A monomer is a molecule that may bind chemically to other molecules to form a polymer. • A polymer is a large molecule (macromolecule) composed of repeating structural units. • Proteins and some carbohydrates are polymers. • Dehydration reactions— smaller molecules bond as water is given off • Hydrolysis reactions— bonds are broken as water is added Control Positive control—goes through all the steps of the experiment and does contain the substance being tested (positive results are expected) Negative control—goes through all the steps of the experiment except it does not contain the substance being tested (negative results are expected) • The control samples usually validate the experiment. If negative control samples give positive results, the experiment is invalid—the reagents may be contaminated or the procedure may need improvement. Carbohydrate • Carbohydrate— sugars and chains of sugars • Glucose— monomer – monosaccharide • Maltose— 2 sugar unit – disaccharide • Glycogen, Starch ect.—chains of glucose units—polysaccharides Tests for carbohydrates • Test for starch (page29) if starch is present Iodine— brown →blue; if starch is absent Iodine— no color change Which tube is the control? • Test for sugars (page31) Benedict’s reagent reacts with small sugar molecules (glucose, maltose etc.) after heating — blue → green/yellow/red !! Highly corrosive Which tube is the control? Protein • Amino acid (Aa) • ~20 different common amino acids • >10 Aa = polypeptide amino group (-H N) acidic group (-COOH) • >50 Aa = protein • antibodies, transport proteins (hemoglobin, albumin), regulatory proteins (insulin); structural proteins (keratin— hair, myosin—muscle); enzyme (speed chemical reactions) 2 Test for protein Biuret reagent (NaOH+CuSO4 ) page 33 Peptide bonds combine with the copper ions. blue→ purple !! Highly corrosive Which tube is the control? Lipids Emulsification of Lipids • Emulsifier can cause a fat to disperse in water Today’s tests • • • • • • • • • • Test for Fat Page 35 15 min Chemical composition unknown A and B Test for sugars Page 31 5-10 min Chemical composition unknown A and B Emulsification of Lipids Page 36 5 min Test for proteins Page 33 Chemical composition unknown A and B Test for starch Page 29 Chemical composition unknown A and B Microscopic observation* Page 30 Page 38 Page 38 Page 38 Page 38