Aldehydes and Ketones
advertisement

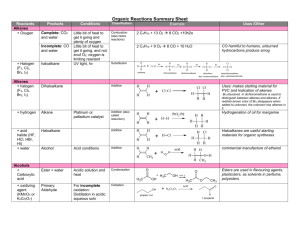
Aldehydes and Ketones • The functional group in aldehydes and ketones is the carbonyl group. O R1 R2 or H carbonyl group Aldehydes and Ketones • Except for formaldehyde, aldehydes have one H atom and one organic group bonded to a carbonyl group. O H3C O O H ethanal or acetaldehyde H H methanal or formaldehyde H 3C C H2 H propanal or propionaldehyde • Ketones have two organic groups bonded to a carbonyl group. H3C O O O C C C CH 3 propanone or acetone H2C CH 3 CH 3 2-butanone or ethylmethylketone H 2C CH 3 CH 2 CH 3 3-pentanone or diethylketone Aldehydes and Ketones • Common names for aldehydes are derived from the name of the acid with the same number of C atoms. • IUPAC names are derived from the parent hydrocarbon name by replacing -e with -al. H3C C H2 H2 C O C H2 C pentanal or pentyl aldehyde H3C H H3C C O O C C H H CH3 2,3-dimethylproponal or 2,3-dimethylpropionaldehyde benzanal or benzyl aldehyde Aldehydes and Ketones • The IUPAC name for a ketone is the characteristic stem for the parent hydrocarbon plus the suffix -one. • A numeric prefix indicates the position of the carbonyl group in a chain or on a ring. O H3C C H2 H2 C O C H2 C CH3 2-hexanone or methyl pentyl ketone H3C H2 C O C H2 C C H2 C CH3 CH3 3-hexanone or ethyl propyl ketone acetophenone or methyl phenyl ketone Amines • Amines are derivatives of ammonia in which one or more H atoms have been replaced by organic groups (aliphatic or aromatic or a mixture of both). • There are three classes of amines. H N H H ammonia H3C N H primary amine H H3C N H CH3 secondary amine H3C N CH3 CH3 tertiary amine Carboxylic Acids O • Carboxylic acids contain the carboxyl functional group. C OH O • The general formula for carboxylic acids is: – R represents an alkyl or an aryl group C R1 OH • IUPAC names for a carboxylic acid are derived from the name of the parent hydrocarbon. – The final -e is dropped from the name of the parent hydrocarbon – The suffix -oic is added followed by the word acid. • Many organic acids are called by their common (trivial) names which are derived from Greek or Latin. Carboxylic Acids O O C C H H3C OH methanoic acid or formic acid ethanoic acid or acetic acid O O H3C H2 C C C H2 OH OH propanoic acid or propionic acid H3C C C H2 OH butanoic acid or butyric acid Carboxylic Acids • Positions of substituents on carboxylic acid chains are indicated by numeric prefixes as in other compounds – Begin the counting scheme from the carboxyl group carbon atom. • They are also often indicated by lower case Greek letters. – = 1st C atom – = 2nd C atom – = 3rd C atom, etc. O H3C H C C OH CH3 CH3 O C C OH H3C H C H2 2-methylpropanoic acid 3-methylbutanoic acid or or -methylpropanoic acid -methylbutanoic acid O H2 H3C H C C C C OH H2 CH3 4-methylpentanoic acid or -methylpentanoic acid Nomenclature of Carboxylic Acids • Dicarboxylic acids contain two carboxyl groups per molecule. O OH C C HO O oxalic acid HO O O O C C C C H2 OH malonic acid HO C H2 H2 C C O succinic acid OH Carboxylic Acids • Aromatic acids are usually called by their common names. • Sometimes, they are named as derivatives of benzoic acid which is considered to be the "parent" aromatic acid. HO HO O benzoic acid HO O O Cl p-chlorobenzoic acid OH O OH CH3 p-toluic acid O phthalic acid Some Derivatives of Carboxylic Acids R1 O O O C C C O R1 acid anhydride R1 acid chloride O O R1 C Cl O ester R2 R1 C NH2 amide When compounds contain more than one functional group, the order of precedence determines which groups are named with prefix or suffix forms. The highest precedence group takes the suffix, with all others taking the prefix form. However, double and triple bonds only take suffix form (-en and -yn) and are used with other suffixes. Functional group Formula Prefix Suffix 1 Cations e.g. Ammonium –NH4+ -onioammonio- -onium -ammonium 2 Carboxylic acids –COOH carboxy- -oic acid* 3 Carboxylic acid derivatives Esters Acyl chlorides Amides –COOR –COCl –CONH2 R-oxycarbonylchloroformylcarbamoyl- -oyl chloride* -amide* 4 Nitrites Isocyanides –CN –NC cyanoisocyano- -nitrile* isocyanide 5 Aldehydes Thioaldehydes –CHO –CHS formylthioformyl- -al* -thial* 6 Ketones Thioketones >CO >CS oxothiono- -one -thione 7 Alcohols Thiols –OH –SH hydroxysulfanyl- -ol -thiol 8 Amines –NH2 amino- -amine 9 Ethers Thioethers –O– –S– -oxy-thio- Priority Isomerism • Isomers have identical composition but different structures • Two forms of isomerism – Constitutional (or structural) – Stereoisomerism • Constitutional – Same empirical formula but different atom-to-atom connections • Stereoisomerism – Same atom-to-atom connections but different arrangement in space. Structural Isomers Stereoisomers: Geometric Geometric isomers can occur when there is a C=C double bond. Cis-2-butene Trans-2-butene Stereoisomers: Optical • Optical isomers are molecules with non-superimposable mirror images. • Such molecules are called CHIRAL • Pairs of chiral molecules are enantiomers. • Chiral molecules in solution can rotate the plane of plane polarized light. • Chirality generally occurs when a C atom has 4 different groups attached. Lactic acid Chirality: Handedness in Nature These molecules are non-superimposable mirror images. Sugars: Related to Alcohols • Sugars are carbohydrates, compounds with the formula Cx(H2O)y. CHO H OH 4 HO HO 5 H HO 3 H H 2 HO 1 OH OH -D-glucose H H H 1 OH 2 H 3 OH 4 OH 5 CH2OH H OH 4 HO HO 5 HO 1 3 H H 2 OH H -D-glucose Open chain form What is the difference between a and b D-glucose? OH Sucrose and Ribose H OH HO HO HO HO H OH O H OH O H H CH2OH -D-Glucose H HO O OH Fructose H H CH2 OH H OH H H H Deoxyribose, the sugar in the DNA backbone. Fats and Oils H2 C HC H2 C O O CR O O CR O O CR What is the functional group in a fat or oil? R = organic group with NO C=C bonds C12 = Lauric acid C16 = Palmitic acid C18 = Stearic acid R = organic group with C=C bonds C18 = oleic acid H2 C HC H2 C O O CR O O CR O O CR Fats and Oils Fats with C=C bonds are usually LIQUDS Oleic acid: a monounsaturated fatty acid C=C bond Trans Fatty Acids •Oleic acid is a mono–unsaturated cisfatty acid •Trans fatty acids have deleterious health effects. •Trans fatty acids raise plasma LDL cholesterol and lower HDL levels. C=C bond Alpha-Amino Acids H2N H O C C OH R Amine H Alanine H3C C Chiral -carbon NH3 CO2 Acid Peptides and Proteins O H 3N OŠ H CH3 Alanine HOCH 2 H 3N + H OŠ O Serine peptide bond – H2O H HOCH2 H H 3N O N H O OŠ CH3 Adding more peptide links ---> PROTEIN Polymers • Giant molecules made by joining many small molecules called monomers • Average production is 150 kg per person annually in the U.S. Polymer Classifications • Thermoplastics (polyethylene) soften and flow when heated • Thermosetting plastics — soft initially but set to solid when heated. Cannot be resoftened. • Other classification: plastics, fibers, elastomers, coatings, adhesives Polymer Preparation • Addition polymers — directly adding monomer units together • Condensation polymers — combining monomer units and splitting out a small water (water) Types of Polyethylene Linear, high density PE (HDPE) Branched, low density PE, LDPE Cross-linked PE, CLPE Types of Polyethylene Polymers based on Substituted Ethylenes, CH2=CHX CH2CH OH CH2CH n polyvinyl alcohol CH2CH OCCH3 n O polyvinyl acetate n polystyrene Table 11.12: others are PVC, acrylonitrile, polypropylene, polymethyl methacrylate Bubble Gum! A copolymer Styrene + butadiene Condensation Polymers O n HOC O COH + n HOCH2CH2OH terephthalic acid O C ethylene glycol O COCH2CH2O + H2 O n Polyethylene terephthalate (PET), a polyester Polyamides: Nylon •Each monomer has 6 C atoms in its chain. •A polyamide link forms on elmination of HCl •Result = nylon 66 •Proteins are polyamides Polymer Recycling Symbols LDPE = HDPE = PP = V= Low density PE = 0.910-0.925 g/cm3 High density PE = 0.941-0.965 Polypropylene = 0.90 PVC (Vinyl chloride) = 1.30-1.58