Science_10-4_files/Science 10-4
advertisement

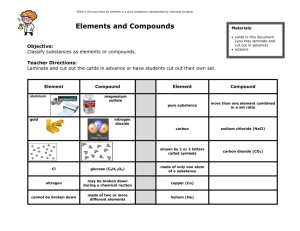
Elements and Compounds Lesson 2 Elements v. Compounds Element – Oxygen Compound – Carbon Dioxide The Letters of Life Just like words are made up of letters, compounds are made up of elements Water Baking Soda Capsaicin Sugar Gun Powder Chips! Questions What is an element? What is a compound? Pure Substance?? Matter Pure Substance Element Compound Mixture Naming Chemicals and Formulas Why would it be confusing if everyone used different names for chemicals? IUPAC International Union of Pure and Applied Chemistry The symbols for the elements were the First Letter of the elements name. When they used that letter the next element stating with the same name used a lower case letter. Periodic Table Questions Why is the Symbol “C” used for Carbon and not for Calcium? Why is the symbol for lead “Pd” and not “Le”? Writing/Reading Chemical Formulas H2O(l) Two Hydrogen One Oxygen Liquid Compound Name Common Use Chemical Formula Calcium carbonate Chalkboard chalk CaCO3(s) Sodium phosphate Heavy-duty cleaner Na3PO4(s) Magnesium chloride De-icing roads MgCl2(s) Monosodium glutamate (MSG) Food seasoning NaC5H8NO4(s) Hydrogen peroxide Bleach and disinfectant H2O2(l) Glucose Sugar C6H12O6(s) Carbon dioxide Fizz in pop, dry ice CO2(g) Freon-12 Refrigerator coolant CCl2F2(g) Number and Name of Elements Total Number of Atoms Decomposition Reaction Water Electrolysis Answer questions 1, 2, 3, 4 on pg 33 H H O Pg. 34 Questions 1-4