Membranes - revised
advertisement

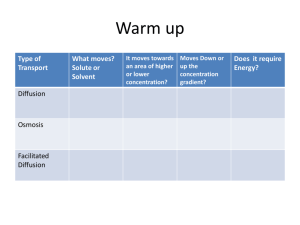
TRANSPORT PROTEINS • Make lipid bilayer permeable to hydrophilic substances: ions and polar molecules • SPAN entire membrane – “transmembrane” • TWO general classes – Channel proteins – Carrier proteins Channel Proteins • A hydrophilic “tunnel” • Example: aquaporins – 3 billion water molecules per second per aquaporin – Water molecules move single file/channel holds 10 – Tremendous increase in rate! Carrier Proteins • Hold on to molecules/ions; change conformation; shuttle across • Specific for substance translocated • Ex. glucose transporter; sodium potassium pump This is a receptor protein • The signal DOES NOT come in • There is NO channel • There is a binding site for the signal. • The message is transmitted to the inside of the cell but THE MESSENGER DOES NOT COME IN Selective Permeability • What determines direction of transport? • What determines whether a substance will enter or leave the cell? • To answer these questions we need to understand 2 modes of transport: – Passive transport – Active transport PASSIVE TRANSPORT ACTIVE TRANSPORT Solutes move DOWN the concentration gradient Solutes move AGAINST the concentration gradient Does not require cell energy – uses the potential energy of the chemical gradient Requires cell energy – ATP directly or indirectly (cotransport) Solutes move between phospholipids or through transport protein (channels or carriers) Solutes bind carriers and carrier changes conformation and translocates solute across the membrane Passive Transport • Passive transport - diffusion of a substance across a biological membrane. • Diffusion - net movement of molecules from high concentration to low concentration (down the concentration gradient) • Results from kinetic energy of molecules. Does not require energy input. • Diffusion continues until a dynamic equilibrium is reached (no net directional movement) • Diffusion – movement of molecules of a substance so that they spread out evenly into the available space • Why do molecules move? • Is the movement here random or directional (net movement)? Explain. • Describe the movement of the molecules at equilibrium. • Is the movement in the pictures “down the concentration gradient” or “against the concentration gradient”? • What is the free energy change for this process? • What is different in this picture? • Where is the concentration gradient highest initially? • Does the diffusion to the purple dye influence the diffusion of the yellow dye? • Would you describe the membrane between the two compartments as permeable to the dye or selectively permeable to the dye? Osmosis • Osmosis - passive transport (diffusion!) of water across a selectively permeable membrane. • What is the role of water in cells? • Biological fluids are usually dilute solutions. • Some water within the cell is not “free” to move down it’s concentration gradient. Why? • Cell membranes are not barriers to diffusion of water. – Aquaporins Osmosis • Relative concentration terms: a) Hypertonic - solution with greater solute concentration than inside the cell. b) Hypotonic - solution with lower solute concentration than inside the cell. c) Isotonic - solution with equal solute concentration compared to that inside a cell. • The prefixes refer to solute concentration. What would the water concentration be in each example above? • What are some units that could be used to express the amount of solute in an aqueous solution? HYPO = LESS HYPER = MORE ISO = EQUAL • TIP! WATER FOLLOWS SOLUTES WHAT ARE THESE THINGS ANYWAY? Movement of Water • If two solutions are separated by a selectively permeable membrane that is permeable to water but not to solute, water will diffuse from the hypoosmotic solution to the hyperoosmotic solution. • “Water moves to DILUTE!” • Direction of osmosis is determined by the difference in total solute concentration, regardless of the types of solutes in the solutions. • At equilibrium, water molecules move in both directions at the same rate. (True for isotonic solutions also) Cell Survival • Balance of water between the cell and its environment are crucial to organisms. • Osmoregulation - control of water balance. • Animal cells (no cell walls) have adaptations for osmoregulation. Ex) Paramecium have contractile vacuoles • Plants can take in water and become turgid (firm), while in isotonic solutions cells become flaccid (limp). In hypertonic solutions, the plant cell shrivels and plasmolysis occurs. • cytolysis plasmolysis Facilitated Diffusion • Diffusion of polar molecules and ions across a membrane with the aid of transport proteins. • Proteins have a specialized binding site for the solute they transport. • Some proteins have gated channels that open or close in response to a stimulus. Facilitated Diffusion Active Transport • Pumping of solutes against the concentration gradient, requiring energy from the cell. • Sodium-potassium pump allows cells to exchange Na+ and K+ across animal cell membranes. • • Ion Pumps • Because anions and cations are unequally distributed across plasma membranes, all cells have voltages across their plasma membrane (membrane potential). • Forces that drive passive transport of ions across membranes include: concentration gradient of the ion (chemical force), and effect of membrane potential (electrical force) on the ion. • The combination of these forces is called the electrochemical gradient. VOLTAGE OCCURS WHEN ELECTICAL CHARGES ARE SEPARATED. Electrogenic Pumps • Transport proteins that generate voltage across a membrane. • Na+/K+ ATPase is the major electrogenic pump in animal cells • A proton pump (H+) is the major electrogenic pump in plants, bacteria, and fungi. • Mitochondria and chloroplasts use a proton pump to drive ATP synthesis. • Voltages created by electrogenic pumps are sources of potential energy available to do cellular work. • Cotransport • Process where a single ATP-powered pump actively transports one solute and indirectly drives the transport of other solutes against their concentration gradients. • Example: Plants use the mechanism of sucrose/H+ cotransport to load sucrose produced by photosynthesis into specialized cells in the veins of leaves. Transport proteins can move sucrose into the cell against the concentration gradient only if it travels with the H+ ion. • Movement of Large Molecules or Cells • Large molecules such as proteins or polysaccharides and cells cross membranes by the processes of endocytosis and exocytosis. • Material to be transported is placed in membrane-bound sacs • Formation and transport of these vesicles requires energy Exocytosis • Process of exporting macromolecules from a cell by fusion of vesicles with the plasma membrane. • Vesicle usually budded from the ER or Golgi and migrates to plasma membrane • Used by secretory cells to export products, such as insulin in the pancreas or neurotransmitters from neurons. Endocytosis • Process of importing macromolecules into a cell forming vesicles derived from the plasma membrane. • Vesicle forms from a localized region of plasma membrane that sinks inward, pinches off into the cytoplasm. • Used by cells to incorporate extracellular substances. Types of Endocytosis: Phagocytosis • Endocytosis of solid particles. • Cell engulfs particle with pseudopodia, and pinches off a food vacuole. • Vacuole fuses with a lysosome containing hydrolytic (digestive) enzymes that break down the particle. • Ex. unicellular protists like amoeba and some immune system cells Phagocytosis of a bacterial cell by an amoeba Types of Endocytosis: Pinocytosis • Endocytosis of fluid droplets • Extracellular fluid is engulfed in small vesicles • Nonspecific in the substances it transports. Cell takes in all solutes dissolved in the droplet. Pinocytosis of fluid into cell lining a blood vessel Types of Endocytosis: Receptor-mediated Endocytosis • Very specific – molecule to be transported (ligand) must bind to receptor of cell surface • After ingested material is released from the vesicle for metabolism, the receptors are recycled to the plasma membrane. • Ex. uptake of cholesterol • Receptor-mediated endocytosis