09.Chapter9.Aldehydes and ketones_1_20121205095814
advertisement

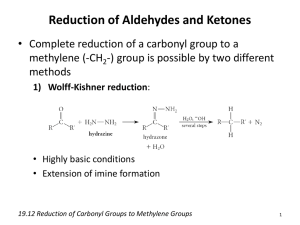
Organic Chemistry Chapter 9 Part I Carbonyl Group (I) Aldehydes and Ketones Nanoplasmonic Research Group Carbonyl Group (I) • Carbonyl Compounds • The alpha hydrogens of a carbonyl compounds are much more acidic than a typical C-H bond. Carbonyl Group (II) • Oxygen is more electronegative than carbon. Therefore, the carbonyl carbon becomes electrophilic, and thus more reactive with nucleophiles. The oxygen can react with an electrophile such as a proton and other Lewis acids Nomenclature of Aldehydes • Acyclic aliphatic aldehydes are named as derivatives of the longest chain containing the aldehyde group by changing the suffix – e of the parent alkane to –al. • -CHO group is attached to a ring, the suffix –carbaldehyde may be used. methanal (formaldehyde) ethanal (actaldehyde) propanal (propionaldehyde) butanal n-butyraldehyde Nomenclature of Ketones • Ketones are named using IUPAC nomenclature by changing the suffix –e of the parent alkane to –one. 2 propanone (acetone) 3 2-butanone (ethyl methyl ketone) 3-pentanone (diethyl ketone) 1 2 3 cyclohexanone 2-metjhylcyclopentanone 2 3-buten-2-one (methyl vinyl ketone) Synthesis of Aldehydes and Ketones • Aldehydes can be prepared by reacting a primary alcohol with an oxidizing agent (an chromium (VI) oxidizing agent) • Ketones can be created by oxidation of secondary alcohol oxidizing agent Reactions of Aldehydes and Ketones • Let’s focus on the reactivity of the carbonyl group!!! • Nucleophilic Addition • Ketones are less reactive than aldehydes • Reduction and Oxidation • Keto-Enol Tautomerism • The Aldol Condensation Nucleophilic Addition to Carbonyl Groups (oxygen (I)) • Addition of Alcohols: Formation of Hemiacetals and Acetals Nucleophilic Addition to Carbonyl Groups (oxygen (II)) • Formation of a cyclic hemiacetal: Generally unstable compounds, in some cases, however, stable cyclic hemiacetals can be readily formed when 5- and 6-membered rings are possible. • Acetal are used as a protecting group for carbonyl groups since they are stable with respect to hydrolysis by bases. Nucleophilic Addition to Carbonyl Groups (carbon (I)) • Nucleophilic Carbon: Grignard Reagents • Nucleophilic Carbon: Acetylides • A metal acetylide is an alkyne for which the terminal proton has been replaced by a metal such as sodium or an organolithium Nucleophilic Addition to Carbonyl Groups (carbon (II)) • Nucleophilic Carbon: Addtion of Hydrogen Cyanide • Hydrogen cyanide has no lone pair electron on carbon. The base can converts to cyanide ions which then can act as a carbon nucleophile since it is weakly acidic. Nucleophilic Addition to Carbonyl Groups (nitrogen) • The tetrahedral addtion of ammonia and amines: similar to a hemiacetal • These addition products are unstable. They can readily eliminate water to form imines!!! -HOH primary amine tetrahedral Addition product imine