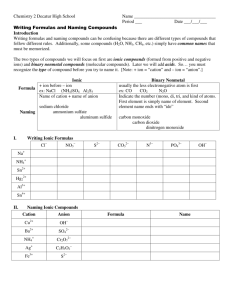
Binary Compounds
Containing Two Nonmetals
To name these compounds, give the name of the less electronegative
element first with the Greek prefix indicating the number of atoms of that
element present, followed by the name of the more electronegative nonmetal with the Greek prefix indicating the number of atoms of that element
present and with its ending replaced by the suffix –ide.
Prefixes you should know:
Mono
Di
Tri
Tetra
Penta
Hexa
Hepta
Octa
Nona
Deca
1
2
3
4
5
6
7
8
9
10
Binary Compounds
Containing Two Nonmetals (Type III Compounds)
As2S3
1. ________________
diarsenic trisulfide
SO2
2. ________________
sulfur dioxide
P2O5
diphosphorus pentoxide
____________________
CO2
4. ________________
carbon dioxide
3.
5.
N2O5
dinitrogen pentoxide
____________________
6.
H2O
dihydrogen monoxide
____________________
Prefixes – Binary
Molecular
Compounds
Greek Prefixes
for
Two
Nonmetals
Number Indicated
1
2
3
4
5
6
7
8
9
10
Prefixes
monoditritetrapentahexaheptaoctanonadeca-
Binary Molecular Compounds
N2O
N2O3
N2O5
dinitrogen monoxide
dinitrogen trioxide
dinitrogen pentoxide
ICl
ICl3
iodine monochloride
iodine trichloride
SO2
SO3
sulfur dioxide
sulfur trioxide
Naming Binary Compounds
Binary Compound?
Yes
Metal Present?
No
Type III
Use Greek
Prefixes
Yes
Does the metal form
more than one cation?
No
Type I
Use the element
name for the cation.
Zumdahl, Zumdahl, DeCoste, World of Chemistry 2002, page 98
Yes
Type II
Determine the charge
of the cation; use a Roman
numeral after the cation
name.
 0
0