6CO 2 - Life Learning Cloud
advertisement

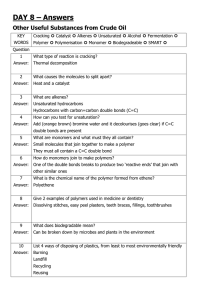
Revision sheets C1 element compound Mixture= 2 or more different atoms not bonded Periodic table 1 Period 1 Period 2 3 4 5 6 3. Test for CO2 Calcium hydroxide reacts with CO2 To make calcium carbonate which Makes limewater go cloudy 2.Quicklime + water slaked lime Calcium oxide + water calcium hydroxide CaO + H2 O Ca(OH)2 5.Cement= limestone + clay + heat Limestone found At quarries-explosives used Causes-noise/air pollution Scars landscape Quarrying good for Local economy/jobs tourism 6. concrete= cement+ gravel+sand+water 7. glass= limestone+sand+ sodium carbonate+heat Extraction methods Iron ore= haematite An alloy is a mixture of metal and other elements Properties that can changeStrength, appearance, hardness, Resistance to corrosion When heated Properties.. • • • • • Good conductors of heat/electricity Strong/hard Dense/heavy Malleable(bent into shape) High melting points Blast furnace Extraction of iron Copper Used in water pipes As doesn’t corrode Used as electrical wires As good conductor Titanium/aluminium Very useful because Strong but lightweight Resist corrosion Carbon reduces iron Oxide to iron Usually recycle aluminium to save money This works because, each fraction has a different Boiling point Crude oil Formula= CnH2n +2 Some fractions take a long time to cool, so rise to top Ease at which It turns into gas Clear colour High volatility 1 = complete combustion 2 = incomplete combustion Low volatility Dark colour (Viscous – gooey, sticky (syrup is viscous)) Alkane names=methane, ethane, propane, butane Pentane, hexane, heptane, octane, nonane, decane New fuels- ethanol-Made from plants-renewable-Carbon neutral-as takes-in as much CO2 in-photosynthesis As it gives-out-when burnt cracking Large alkanes are not very useful to use, so they are broken down into smaller ones, like petrol, using a process called cracking. Cracking requires a catalyst so the process is called catalytic cracking. Alkanes are saturated they have no carbon to carbon double bonds. Thermosoftening plastics C10H22 C2H4 + C3H8 + C5H10 e.g of cracking reaction The products must either be alkanes (have a formula of CnH2n+2 or alkenes CnH2n) Test for unsaturated alkenes. Add bromine water. it will change From yellow/orange to colourless Alkenes are unsaturated. They have carbon to carbon double bonds. There are weak intermolecular forces between the plastics chains. These plastics are soft and melt when heated. An example is low density polythene (LDPE). They Disposal of polymers are used to make plastic bags. 1 landfill sites- fill up as plastics usually not biodegradable 2. burning- releases CO2- leads to global warming 3. recycling- best option as saves money, energy Thermosetting plastics There are crosslinks between the chains. This means that the intermolecular forces are strong. Thermosetting plastics are hard and do not melt. An example of a thermosetting plastic is high density polythene (HDPE). Use= buckets We can make polymers using alkenes. The alkene molecule (for example ethane) is called a monomer (mono means 1, mer means part). It makes up 1 part of the plastic polymerisation When alkenes are turned into polymers, their carbon to carbon double bond breaks When these monomers are bonded together in a huge chain, they are called polymers (poly means many, mer means part.) Biofuels Renewable- can be replaced /regrown Do not add any more CO2 to atmosphere Carbon neutral No sulphur dioxide produced More biodegradable than diesel Plants produce glucose using photosynthesis…..some glucose changed to oils 6CO2 (carbon dioxide) + 6H2O (water) C6H12O6 (glucose) + 6O2 (oxygen) We can saturate vegetable oils using a process called hydrogenation. Unsaturated oils are passed over a nickel catalyst with hydrogen at 60oC. This process is used to make margarine. We can also said that the oils have been hardened. It is an addition reaction Oil and water don’t mix. They are immiscible. Substances that do mix are miscible. We have an emulsion when we have small droplets of oil mixed in with water. However, since oil and water don’t mix, eventually, this emulsion separates again. E.g include ice-cream, milk, mayo, sauces We keep oil and water mixed by adding an emulsifier. Emulsifiers have a part that mixes with water and a part that mixes with oil. Egg yolk is an emulsifier Oil droplets remain separate Vegetable oils Full of energy Extracted by crushing, pressing Or distillation Chromatography( use to separate colours) Chromatography is used to separate substances. It works because different substances dissolve better in water than others. The more soluble they are in the solvent, the further they travel up the paper A and C contain the same 2 dyes B contains 3 dyes D contains at least 1 insoluble dye E100s range – colours additives E200s range – preservatives. Keeps food longer E300s range – Antioxidants. Stops food reacting with oxygen E400s range – Emulsifiers, stabiliers and thickeners E500s range – Acidity regulators E600s range - Flavourings Almost solid, but flows slowly liquid solid Lithosphere Dense Iron and nickel Pangea 1 massive continent Used in light bulbs All noble gases: Are gases at room temperature Exist as single atoms (monatomic) Do not react with anything Plate tectonics This is because the Earth’s crust and Helium, Neon, Argon, Krypton upper mantle are split into tectonic Xenon, Radon plates. These plates travel along Group 8/0 of periodic table convection currents caused by heating Neon – Used in lighting, from radioactive materials. This caused glows with high voltage continental drift. Evidence of continental drift atmosphere Fault lines Initially thought They where formed By the shrinking of The earth’s crust volcano mountain Earthquake ( occur randomly on fault lines) If earthquakes form under the ocean, then it will form a tidal wave called a tsunami. • • Continents fit together Same fossils found on each continent Atmosphere before 18/20% Atmosphere now 78/80%